Arq. Bras. Oftalmol. 2026; 89 (3): 10.5935/0004-2749.2025-0312
Total: 501
Isabella Passarelli Giabardo Marques1; Christiana Velloso Rebello Hilgert2,3; Caio Henrique Peres Oliani1; Juliana Carolina Contrera1; Marcello Novoa Colombo-Barboza1,4; Guilherme Novoa Colombo-Barboza1,5; Bernardo Kaplan Moscovici1,3
DOI: 10.5935/0004-2749.2025-0312
ABSTRACT
PURPOSE: To quantitatively assess changes in central corneal sensitivity after phacoemulsification and to characterize recovery patterns up to 90 days using standardized esthesiometry.
METHODS: This prospective observational study included 44 patients (88 eyes) undergoing uncomplicated phacoemulsification with intraocular lens implantation. Central corneal sensitivity was measured using a Cochet-Bonnet® esthesiometer preoperatively and at 30 and 90 days postoperatively. Repeated-measures data were analyzed using Friedman and Wilcoxon signed-rank tests (p<0.05). Inter-eye differences were assessed with a paired Wilcoxon test. Individual changes from baseline (Δ30, Δ90) were calculated, and 90-day recovery was categorized according to thresholds aligned with the 5-mm device resolution. Spearman correlation was used to explore associations between age and Δ90.
RESULTS: Corneal sensitivity decreased after surgery. In right eyes, mean sensitivity declined from 41.14 ± 7.77 mm at baseline to 36.82 ± 9.03 mm at 30 days and partially recovered to 38.64 ± 7.73 mm at 90 days. In left eyes, sensitivity decreased from 44.11 ± 6.29 mm to 37.39 ± 9.05 mm at 30 days and recovered to 41.82 ± 7.63 mm at 90 days. Left eyes showed higher sensitivity than right eyes at baseline (p=0.023) and at 90 days (p=0.018). At 90 days, complete or near-complete recovery (within ± 5 mm of baseline) occurred in 73.2% of right eyes and 78.0% of left eyes, while improvement above baseline (≥ +5 mm) occurred in 7.3% and 4.9%, respectively. Age showed weak, nonsignificant correlations with Δ90 (p=−0.14 to −0.19; p>0.2).
CONCLUSION: Phacoemulsification with a 2.75-mm clear corneal incision leads to a temporary reduction in central corneal sensitivity, with partial recovery by 90 days. Recovery patterns vary among individuals, highlighting the value of postoperative sensitivity monitoring to identify atypical trajectories and guide ocular surface care during visual rehabilitation.
Keywords: Phacoemulsification; Cornea/innervation; Ophthalmic nerve/physiology; Optometry/instrumentation; Diagnostic techniques, ophthalmological; Neural regeneration; Visual rehabilitation.
INTRODUCTION
The cornea is an avascular, transparent, and densely innervated structure of the eye, contributing approximately two-thirds of its refractive power. Its anatomical and functional integrity is essential for maintaining visual acuity and protecting the eye from mechanical, thermal, and chemical insults. Corneal sensitivity is mediated by a complex network of sensory nerve fibers originating from the ophthalmic branch of the trigeminal nerve, which form intraepithelial, subepithelial, and stromal plexuses. These nerves play a critical role in detecting harmful stimuli; triggering protective reflexes, such as blinking and tearing; and maintaining ocular surface homeostasis, including epithelial wound healing and regulation of the tear film(1).
With an aging global population, cataract has become one of the leading causes of preventable visual loss worldwide. Characterized by progressive opacification of the crystalline lens, cataracts impair lens transparency and interfere with light transmission to the retina, leading to reduced visual acuity, photophobia, blurred vision, and, in advanced cases, functional blindness(2). Globally, more than 50% of cases of reversible blindness are attributable to cataract, underscoring the importance of early detection and timely surgical intervention(3).
Phacoemulsification is currently the gold standard for surgical cataract treatment, owing to its safety, effectiveness, minimal invasiveness, and rapid postoperative recovery. The procedure involves ultrasonic fragmentation of the opacified lens, followed by implantation of an intraocular lens. Despite technological advances, phacoemulsification requires small corneal incisions that may transiently disrupt corneal nerve endings, resulting in temporary alterations in corneal sensitivity(4-11).
Corneal hypoesthesia in the immediate postoperative period following phacoemulsification is well documented. Significant decreases in central corneal sensitivity can be detected as early as the third postoperative day, with incomplete recovery often observed even after 3 months. Similar studies have reported marked reductions in both central and temporal corneal sensitivity, followed by gradual improvement over the first 3 months after surgery. The extent of recovery may be influenced by factors such as the location and size of the corneal incisions, patient age, individual inflammatory response, and comorbidities, including diabetes mellitus(11-17).
This study prospectively and quantitatively investigates changes in corneal sensitivity following phacoemulsification, with a particular focus on the time required for recovery to preoperative levels. By employing esthesiometry as an objective measurement tool, the study aims to enhance understanding of the sensory effects of cataract surgery and provide evidence to inform postoperative management, ultimately supporting improved visual outcomes and patient comfort.
METHODS
Study design and population
This prospective observational study included 44 patients (88 eyes) who underwent uncomplicated phacoemulsification with intraocular lens implantation at a tertiary ophthalmology center. Both eyes of each participant were evaluated, and data were analyzed as paired observations where appropriate. All participants provided written informed consent prior to enrollment. The study was conduct in accordance with the principles of the Declaration of Helsinki and was approved by the institutional ethics committee.
Inclusion and exclusion criteria
Eligible participants were adults (≥18 years) with a clinical indication for cataract surgery who were able to comply with follow-up visits and study assessments. Exclusion criteria included pre-existing corneal pathology (e.g., keratitis, dystrophies, or degenerations), a history of prior ocular surgery, contact lens use within 3 months before surgery, and systemic conditions that could affect corneal innervation, particularly uncontrolled diabetes mellitus. Cases with intraoperative complications, such as posterior capsular rupture or zonular dialysis, were also excluded from the analysis.
Surgical procedure
All surgeries were performed by a single experienced surgeon using a standardized phacoemulsification technique under topical anesthesia. A temporal clear corneal incision of 2.75 mm was created, followed by continuous curvilinear capsulorhexis, hydrodissection, phacoemulsification using the chop technique, cortical aspiration, and implantation of a foldable acrylic posterior chamber intraocular lens into the capsular bag. All procedures included in the study were uneventful. Operative duration and cumulative dissipated ultrasound energy were not recorded for the present analysis.
Corneal sensitivity assessment
Central corneal sensitivity was measured using a Cochet-Bonnet® esthesiometer (Luneau Ophthalmologie, France) at three time points: preoperatively (baseline) and at 30 and 90 days postoperatively. The instrument's nylon monofilament was initially set to 60 mm and progressively shortened in 5-mm increments until the participant reported tactile perception. The filament length corresponding to the first positive response was recorded as the corneal sensitivity threshold. All measurements were performed exclusively in the central cornea (within a 4-mm zone) to ensure reproducibility and consistency across visits. Results are expressed in millimeters (mm), consistent with the device scale, where longer filament lengths indicate higher corneal sensitivity (i.e., a lower mechanical threshold). All assessments were conducted by trained personnel under controlled room conditions, with standardized lighting and patient positioning, to minimize variability.
Data collection and statistical analysis
Data were compiled using Microsoft Excel® and analyzed with SPSS version 26 (IBM Corp., Armonk, NY, USA). Given the repeated-measures design and the nonparametric distribution of corneal sensitivity values, nonparametric tests were employed. The Friedman test was used to evaluate differences across the three time points, and when significant differences were identified, pairwise comparisons were performed using the Wilcoxon signed-rank test. Values are reported as mean ± standard deviation (SD), median, and interquartile range to describe central tendency and dispersion. Non-normal distribution was confirmed by the Shapiro–Wilk test and visual inspection of histograms; accordingly, nonparametric tests were applied to all analyses. A p-value <0.05 was considered statistically significant.
Inter-eye differences were evaluated using paired Wilcoxon signed-rank tests to compare right and left eyes at each time point.
Individual changes from baseline were calculated as Δ30 (postoperative day 30 minus baseline) and Δ90 (postoperative day 90 minus baseline) and were visualized using trajectory plots and distribution analyses.
To characterize clinical heterogeneity in recovery at 90 days, eyes were categorized based on Δ90 relative to baseline according to thresholds aligned with the 5-mm resolution of the Cochet-Bonnet® esthesiometer: improvement above baseline (Δ90 ≥ +5 mm), complete or near-complete recovery (Δ90 within ± 5 mm), partial recovery (Δ90 between −6 and −10 mm), and no recovery or worsening (Δ90 < −10 mm or ≤ value at Δ30). Proportions in each category were reported separately for right and left eyes.
An exploratory Spearman rank correlation was used to assess the relationship between patient age and sensory recovery (Δ90).
RESULTS
A total of 44 patients (88 eyes) were included in the study, with both eyes evaluated in all participants. The mean age was 67.9 years (SD 8.8; median 67.5 years; range 36–86 years).
Central corneal sensitivity values at each time point are summarized in table 1 and illustrated in figure 1. The Friedman test demonstrated statistically significant differences across the three time points for right eyes (p=0.002), left eyes (p<0.001), and both eyes combined (p<0.001). Post hoc pairwise comparisons, performed using the Wilcoxon signed-rank test, are presented in table 2.
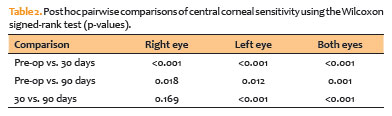
In the right eyes, mean central corneal sensitivity decreased from 41.14 ± 7.77 mm preoperatively to 36.82 ± 9.03 mm at 30 days postoperatively (PO-30), with partial recovery to 38.64 ± 7.73 mm at 90 days postoperatively (PO-90). The decline from baseline was statistically significant at both PO-30 (p<0.001) and PO-90 (p=0.018), whereas the difference between PO-30 and PO-90 was not significant (p=0.169).
In the left eyes, sensitivity decreased from 44.11 ± 6.29 mm at baseline to 37.39 ± 9.05 mm at PO-30, recovering to 41.82 ± 7.63 mm at PO-90. All pairwise comparisons were statistically significant (preoperative vs. PO-30: p<0.001; preoperative vs. PO-90: p=0.012; PO-30 vs. PO-90: p<0.001).
When both eyes were analyzed together, mean sensitivity declined from 42.63 ± 7.18 mm preoperatively to 37.10 ± 8.99 mm at PO-30 and partially recovered to 40.23 ± 7.80 mm at PO-90, with all pairwise comparisons statistically significant (Table 2).
Paired inter-eye comparisons (Table 3) indicated that left eyes had higher sensitivity than right eyes at baseline (mean difference +2.97 mm, p=0.023) and at PO-90 (mean difference +3.18 mm, p=0.018). In contrast, no significant difference was observed at PO-30 (mean difference +0.57 mm, p=0.663).
Individual trajectory analysis (Figure 2) revealed substantial intersubject variability in both the magnitude and pattern of sensory changes. Most eyes exhibited a marked reduction in sensitivity at PO-30, followed by variable degrees of recovery at PO-90 (Figure 2A, right eyes; Figure 2B, left eyes).
The distribution of individual changes from baseline (Figure 3) confirmed a consistent decrease at PO-30 and partial recovery at PO-90, with greater variability observed in the left eyes.
Recovery patterns at 90 days relative to baseline are summarized in table 4 and figure 4. Using categories aligned with the 5-mm resolution of the Cochet-Bonnet® esthesiometer, the majority of eyes achieved complete or near-complete recovery (within ± 5 mm of baseline): 73.2% of right eyes and 78.0% of left eyes. Improvement above baseline (≥ +5 mm) occurred in 7.3% of right eyes and 4.9% of left eyes. Partial recovery (>5 mm below baseline) was observed in 12.2% of eyes in both groups, whereas no recovery or worsening was noted in 7.3% of right eyes and 4.9% of left eyes.
Exploratory Spearman rank correlations between patient age and sensory recovery at 90 days (Δ90) revealed weak negative associations (right eyes: p=−0.14, p=0.39; left eyes: p=−0.19, p=0.22), which were not statistically significant.
DISCUSSION
This prospective study quantitatively demonstrated a significant decline in central corneal sensitivity following phacoemulsification, with partial recovery observed 90 days postoperatively. In the right eyes, mean sensitivity decreased from 41.14 mm preoperatively to 36.82 mm at 30 days and then increased to 38.64 mm at 90 days, with a similar pattern observed in the left eyes. Considerable interindividual variability was noted in postoperative changes, with most eyes showing improvement by 90 days, while a smaller subset exhibited persistent reductions or values exceeding baseline(18-30). These results are consistent with previous studies reporting the transient but measurable effects of cataract surgery on corneal innervation and the gradual activation of neural regenerative mechanisms(10-17).
The anatomical basis for postoperative corneal hypoesthesia lies in the subbasal nerve plexus, which consists of thin, highly branched fibers that traverse Bowman's layer and the anterior stroma, forming a dense network that is readily disrupted by surgical trauma(1,12-16). Although phacoemulsification is considered minimally invasive, mechanical stress from the corneal incision, ultrasonic energy, and irrigating fluids can cause partial denervation and local inflammation, resulting in hypoesthesia and altered ocular surface homeostasis(6,10,17).
Although the temporal clear corneal incision (2.75 mm) directly transects peripheral nerve trunks at the wound site, this study assessed only central corneal sensitivity; therefore, the results reflect global central sensory changes rather than incision-site denervation. Central hypoesthesia may arise from diffuse mechanical stress (e.g., fluid turbulence and intraocular manipulation), postoperative inflammation, and broader sensory modulation, potentially underestimating localized peripheral deficits or regional differences near the incision. Contralateral sensory effects following unilateral corneal injury have also been reported, suggesting that postoperative sensory changes may not be entirely localized(22-30).
Beyond the incision, other aspects of phacoemulsification may contribute to reduced corneal sensitivity. Irrigation and aspiration flow, ultrasonic energy, intraoperative manipulation, and postoperative inflammation can impose mechanical, thermal, and osmotic stress across the cornea, including transient edema, resulting in a diffuse impact on corneal nerves and ocular surface homeostasis. These observations support interpreting the postoperative decline as the consequence of total corneal trauma rather than incision-related nerve transection alone(22-30).
Recovery of corneal sensory function involves axonal regeneration, Schwann cell activation, and synaptic reconnection. This process can be detected as early as 4 weeks postoperatively but often remains incomplete after several months(13,14,18). In vivo confocal microscopy (IVCM) studies have demonstrated that nerve fiber density gradually increases after surgery, correlating with functional recovery assessed by esthesiometry(17,18). Considerable variability in regenerative capacity is expected, with factors such as patient age, inflammatory response, and baseline nerve status influencing outcomes(14,16,18-22). Futures combining esthesiometry with structural and inflammatory markers—such as IVCM-derived nerve metrics and inflammatory cell assessment—could further elucidate the mechanisms underlying incomplete recovery(22-30).
Surgical parameters, particularly incision size and location, influence the extent of corneal neural injury. Microincisional techniques (≤2.75 mm) are associated with fewer nerve transections, faster recovery, and reduced postoperative discomfort compared with larger incisions(4,6,11,19-30). In the present study, the use of a 2.75-mm temporal clear corneal incision likely contributed to the sensory profile observed at 90 days. Furthermore, even unilateral procedures may induce bilateral reductions in corneal nerve density, potentially via systemic or reflex mechanisms(20). These observations support central corneal sensitivity as an integrated outcome influenced by both local and nonlocal factors(22-30).
Age is a plausible modifier of recovery, as neural regeneration tends to be slower in older individuals(30). Procedural duration and other phacoemulsification parameters may also influence outcomes, as longer surgeries can reflect greater intraoperative manipulation and mechanical stress; these variables should be captured in future studies(22-30).
An inter-eye asymmetry in sensitivity was observed, with left eyes exhibiting higher sensitivity than right eyes at both baseline and 90 days. While speculative, this difference may reflect anatomical variations in innervation, ocular dominance, or differential postoperative inflammatory responses(12,14,16). The absence of significant asymmetry at 30 days suggests similar acute injury, with divergence over time reflecting individual recovery trajectories(13,17). Other contributing factors may include surgeon ergonomics and handedness, as well as contralateral neuroimmune modulation reported after unilateral corneal insult(30).
Corneal sensitivity is critical for maintaining ocular surface integrity, regulating blink reflexes, tear secretion, epithelial homeostasis, and nociceptive signaling. Even transient denervation can predispose patients to dry eye symptoms, neurotrophic keratitis, or neuropathic ocular pain(5,8,10,21). Recent insights into dry eye pathophysiology highlight that corneal nerve dysfunction plays a central role in disease progression and symptom severity(18-30). Functional assessment using esthesiometry, particularly when combined with IVCM, can therefore be valuable for postoperative monitoring(13,14,17).
Comorbid systemic conditions, especially diabetes mellitus, further complicate corneal regeneration. Diabetic patients exhibit reduced corneal nerve density and sensitivity at baseline, with delayed or incomplete recovery following surgery[7–9,14]. Although individuals with diabetes were excluded from this cohort, these considerations remain clinically important, emphasizing the need for tailored postoperative care in at-risk populations.
Technological advances have expanded the assessment of corneal innervation. Although the Cochet-Bonnet® esthesiometer is practical, it has limitations, including subjectivity and limited resolution. In contrast, IVCM provides detailed morphological visualization of corneal nerves and can detect regenerative changes and features of nerve regrowth(14,17). When used together, these tools offer complementary information. Cochet-Bonnet® measurements are stepwise (5-mm increments) and subject to ceiling and floor effects, as well as repeatability constraints, which may limit the detection of subtle change(22-30).
The therapeutic implications of corneal nerve impairment are increasingly recognized. Beyond artificial tears and anti-inflammatory agents, neuroprotective strategies are being investigated. Recombinant human nerve growth factor (rhNGF) promotes epithelial healing and improves corneal sensitivity in neurotrophic keratitis and may have potential relevance for postsurgical recovery. Additional approaches under investigation include antiapoptotic peptides, topical insulin, and regenerative scaffolds(14,19).
The present study underscores the value of longitudinal monitoring following cataract surgery. Single measurements may fail to detect delayed recovery, whereas follow-up assessments can identify persistent sensory impairment and guide timely intervention(14,18). Incorporating corneal sensitivity testing may also reveal symptoms not captured by visual acuity alone, including dryness, discomfort, and neuropathic-like ocular pain(5,21). Such complaints likely reflect impaired corneal nerve regeneration and disruption of the corneal–lacrimal reflex pathway.
Several limitations of this study should be acknowledged. Follow-up was limited to 90 days, which may underestimate the time required for full sensory recovery. Only central corneal sensitivity was assessed, without evaluation of regional variations near the incision. A single esthesiometry method was employed, without structural correlation from nerve imaging. Stratified analyses by age or ocular dominance were not performed. Additional limitations include the absence of objective surgical "load" metrics, such as operative duration and ultrasound energy, and the lack of inflammatory or structural correlation (e.g., via IVCM) to distinguish focal incision-related injury from diffuse postoperative inflammation or neuroimmune modulation(22-30).
In conclusion, phacoemulsification with a 2.75-mm clear corneal incision produces a measurable decline in central corneal sensitivity, with partial recovery observed by 90 days. These findings highlight the neural impact of cataract surgery and support the potential incorporation of corneal sensitivity testing into postoperative evaluation. Future studies should extend follow-up periods, assess peripheral and peri-incisional sensitivity, and explore neuroprotective interventions. Additionally, integrating structural imaging with functional testing and recording surgical covariates would help distinguish incision-specific denervation from total corneal trauma and postoperative inflammation(14,17–19,21).
ACKNOWLEDGMENTS
The authors thank the surgical and administrative staff of Hospital Visão Laser for their assistance with patient recruitment and follow-up.
AUTHORS' CONTRIBUTIONS
Significant contribution to conception and design: Isabella Passarelli Giabardo Marques; Christiana Velloso Rebello Hilgert; Caio Henrique Peres Oliani; Juliana Carolina Contrera; Marcello Novoa Colombo-Barboza.
Data Acquisition: Isabella Passarelli Giabardo Marques; Christiana Velloso Rebello Hilgert; Caio Henrique Peres Oliani; Juliana Carolina Contrera; Marcello Novoa Colombo-Barboza.
Data Analysis and interpretation: Guilherme Novoa Colombo-Barboza; Bernardo Kaplan Moscovici.
Manuscript Drafting: Isabella Passarelli Giabardo Marques; Christiana Velloso Rebello Hilgert; Caio Henrique Peres Oliani; Juliana Carolina Contrera; Marcello Novoa Colombo-Barboza.
Significant intellectual content revision of the manuscript: Guilherme Novoa Colombo-Barboza; Bernardo Kaplan Moscovici.
Final approval of the submitted manuscript: Isabella Passarelli Giabardo Marques; Christiana Velloso Rebello Hilgert; Caio Henrique Peres Oliani; Juliana Carolina Contrera; Marcello Novoa Colombo-Barboza; Guilherme Novoa Colombo-Barboza; Bernardo Kaplan Moscovici.
Statistical analysis: Guilherme Novoa Colombo-Barboza
Obtaining funding: not applicable.
Supervision of Administrative, technical, or material support: Christiana Velloso Rebello Hilgert; Bernardo Kaplan Moscovici.
Research group leadership: Bernardo Kaplan Moscovici.
REFERENCES
1. Müller LJ, Pels L, Vrensen GFJM. Ultrastructural organization of human corneal nerves. Invest Ophthalmol Vis Sci. 2003;44(2):527–34.
2. Moreau KL, King JA. Protein misfolding and aggregation in cataract disease and prospects for prevention. Trends Mol Med. 2012;18(5):273–82.
3. WHO. World report on vision [Internet] Geneva: WHO; 2021. [cited 2025 Nov 21]. Availabe from World report on vision
4. Al Mahmood AM, Al-Swailem SA, Behrens A. Clear corneal incision in cataract surgery. Middle East Afr J Ophthalmol. 2014;21(1):25–31.
5. Khanal S, Tomlinson A, Esakowitz L, Bhatt P, Jones H, Nabili S, et al. Changes in corneal sensitivity and tear physiology after phacoemulsification. Ophthalmic Physiol Opt. 2008;28(2):127–34.
6. Ku HC, Lee YC, Kim HS. Corneal sensation and tear film stability after cataract surgery with clear temporal corneal incision. J Korean Ophthalmol Soc. 2004;9:2000–6
7. Kallinikos P, Berhanu M, O'Donnell C, Boulton A, Efron N. Tear production and corneal sensitivity in diabetes. Acta Ophthalmol Scand. 2006;84(6):796–800.
8. Rosenberg ME, Tervo TM, Immonen IJ, Müller LJ, Grönhagen-Riska C, Vesaluoma MH. Corneal structure and sensitivity in type 1 diabetes mellitus. Invest Ophthalmol Vis Sci. 2000;41(10):2915-21.
9. Tavakoli M, Kallinikos P, Efron N, Boulton AJ, Malik RA. Corneal sensitivity is reduced and relates to the severity of neuropathy in patients with diabetes. Diabetes Care. 2007;30(7):1895–7. Comment in: Diabetes Care. 2007;30(12):e142; author reply e143.
10. Kim JH, Chung JL, Kang SY, Kim SW, Seo KY. Change in corneal sensitivity and corneal nerve after cataract surgery. Cornea. 2009;28(9):928–34.
11. Gharaee H, Mousavi M, Daneshvar R, Hosseini M, Khosravi A. Effect of incision size on corneal sensitivity after phacoemulsification. Iran J Ophthalmol. 2009;21(3):29–34.
12. Marfurt CF, Cox J, Deek S, Dvorscak L. Anatomy of the human corneal innervation. Exp Eye Res. 2010;90(4):478–92.
13. Lum E, Corbett MC, Murphy PJ. Corneal sensitivity after ocular surgery. Eye Contact Lens. 2019;45(4):226–37.
14. Gad H, Elgassim E, Lebbe A, MacDonaldo RS, Baraka A, Joannis N, et al. Corneal confocal microscopy detects early nerve regeneration after pharmacological and surgical interventions: a systematic review and meta-analysis. J Peripher Nerv Syst. 2024;29(1):173–84.
15. John T. Corneal sensation after small incision, sutureless, one-handed phacoemulsification. J Cataract Refract Surg. 1995;21(4):425–8.
16. Al-Aqaba MA, Dhillon VK, Mohammed I, Said DG, Dua HS. Corneal nerves in health and disease. Prog Retin Eye Res. 2019;73:100762.
17. De Milliano PA, van Velthoven ME, van der Velde EA, Mourits MP, Lapid-Gortzak R. Regeneration of corneal nerves after cataract surgery: a longitudinal in vivo confocal microscopy study. Cornea. 2022;41(9):1050–7.
18. Aggarwal S, Kheirkhah A, Cavalcanti BM, Cruzat A, Jamali A, Dana R. Corneal nerve density and sensitivity in patients with dry eye disease: a systematic review. Ocul Surf. 2021;19:50–61.
19. Bonini S, Lambiase A, Rama P, Sinigaglia F, Allegretti M, Chao W, Mantelli F; REPARO Study Group. Phase II randomized, double-masked, vehicle-controlled trial of recombinant human nerve growth factor for neurotrophic keratitis. Ophthalmology. 2018;125(9):1332-43. Comment in: Ophthalmology. 2019;126(2):e14-e15. Ophthalmologe. 2019;116(11):1006-7.
20. Hamrah P, Cruzat A, Dastjerdi MH, Zheng L, Shahatit BM, Bayhan HA, et al. Unilateral phacoemulsification leads to bilateral reduction in corneal nerve density. Ophthalmology. 2013;120(6):1271–9.
21. Rosenthal P, Borsook D. The corneal pain system. Part I: the missing piece of the dry eye puzzle. Ocul Surf. 2012;10(1):2–14.
22. Belmonte C, Acosta MC, Gallar J. Neural basis of sensation in intact and injured corneas. Exp Eye Res. 2004;78(3):513–25.
23. Guerrero-Moreno A, Baudouin C, Melik Parsadaniantz S, Réaux-Le Goazigo A. Morphological and functional changes of corneal nerves and their contribution to peripheral and central sensory abnormalities. Neurosci. Front Cell Neurosci. 2020;14:610342.
24. Luna C, Quirce S, Aracil-Marco A, Belmonte C, Gallar J, Acosta MC. Unilateral corneal insult also alters sensory nerve activity in the contralateral eye. Front Med (Lausanne). 2021;8:767967.
25. Miller KM, Oetting TA, Tweeten JP, Carter K, Lee BS, Lin S, Nanji AA, Shorstein NH, Musch DC; American Academy of Ophthalmology Preferred Practice Pattern Cataract/Anterior Segment Panel. Cataract in the adult eye preferred practice pattern®. Ophthalmology. 2022;129(1):P1-P126.
26. Sitompul R, Sancoyo GS, Hutauruk JA, Gondhowiardjo TD. Sensitivity change in the cornea and tear layer due to incision difference in cataract surgery with either manual small-incision cataract surgery or phacoemulsification. Cornea. 2008;27(Suppl 1):S13–8.
27. Niktinat H, Alviar M, Kashani M, Massoumi H, Djalilian AR, Jalilian E, et al. Aging and corneal nerve health: mechanisms of degeneration and emerging therapies for the cornea. Cells. 2025;14(21):1730.
28. Chao C, Stapleton F, Badarudin E, Golebiowski B. Ocular surface sensitivity repeatability with Cochet-Bonnet esthesiometer. Optom Vis Sci. 2015;92(2):183–9.
29. Ruiz-Lozano RE, Quiroga-Garza ME, Ramos-Dávila EM, Pantaleón-García J, Khodor A, Komai S, et al. Comparative evaluation of the corneal sensitivity thresholds between the novel noncontact and Cochet-Bonnet esthesiometers. Am J Ophthalmol. 2025;271:407–16.
30. Cruzat A, Qazi Y, Hamrah P. In vivo confocal microscopy of corneal nerves in health and disease. Ocul Surf. 2017;15(1):15–47.
Data Availability Statement: The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request, subject to justified conditions.
Edited by
Edited by Editor-in-Chief: Newton Kara-Júnior
Associate Editor: André Messias
Submitted for publication:
October 14, 2025.
Accepted for publication:
February 10, 2026.
Approved by the following research ethics committee: Irmandade Santa Casa de Misericórdia de Santos – ISCMS (CAAE: 79683824.2.0000.0139).
Funding: This study received no specific financial support.
Disclosure of potential conflicts of interest: The authors declare no potential conflicts of interest.
How to cite this article: