Arq. Bras. Oftalmol. 2026; 89 (3): 10.5935/0004-2749.2025-0259
Total: 458
Camyla Lemos Budib1; Caio Henrique Peres Oliani1; Guilherme de Oliveira Ferreira2; Ulysses Tachibana3; José Álvaro Pereira Gomes3; Bernardo Kaplan Moscovici1,3,4; Luiz Roberto Colombo-Barboza1; Marcello Novoa Colombo-Barboza1,5; Guilherme Novoa Colombo-Barboza1,6
DOI: 10.5935/0004-2749.2025-0259
ABSTRACT
PURPOSE: To evaluate the reliability and comparability of a Scheimpflug-based tomographer relative to a Placido-based topographer and specular microscopy in healthy eyes.
METHODS: This cross-sectional study included 40 patients (80 eyes). Each eye underwent randomized imaging with a Scheimpflug-based tomographer, a Placido-based topographer, and Tomey EM-4000 specular microscopy. Three acquisitions per device were obtained. For interdevice comparisons, the best-quality scan per eye/device was selected, whereas all three scans were used for intradevice repeatability analyses. Unreliable scans were repeated (up to five attempts) and excluded if acceptable quality was not achieved, resulting in variable denominators. Between-device comparisons were performed using generalized estimating equations
with participant-level clustering and robust standard errors and were supplemented by Bland–Altman analysis.
RESULTS: The effective sample size varied by parameter (independent summaries: 59–67 eyes; paired comparisons: 48–51 eyes). In paired-eye analyses, the Scheimpflug-based tomographer measured slightly higher keratometry values than the Placido-based topographer (K1: 43.95 vs. 43.78 D, p=0.003; K2: 44.91 vs 44.73 D, p=0.002), more negative Q-values (p=0.001), smaller photopic pupil diameter (p<0.001), and shorter kappa distance (p<0.001). Mean absolute differences were 0.32 D for K1 and 0.30 D for K2, with high dispersion for angular metrics (kappa angle coefficient of variation: 195%).
CONCLUSIONS: The Scheimpflug-based tomographer provides reproducible corneal measurements in healthy eyes. However, systematic differences relative to the Placido-based topographer—particularly for keratometry, asphericity, and pupil and kappa metrics—suggest limited interchangeability. Consistent device use is recommended when these parameters inform clinical decision-making.
Keywords: Scheimpflug tomography; Placido topography; Specular microscopy; keratometry; Corneal imaging; Refractive surgical procedures; Lenses, intraocular
INTRODUCTION
The cornea contributes nearly 70% of the eye's total refractive power, making precise measurement of its curvature, thickness, and topography essential for clinical diagnosis and surgical planning(1). Corneal imaging is critical for the early detection of ectatic disorders such as keratoconus, for screening candidates for refractive surgery, and for calculating intraocular lens (IOL) power in cataract surgery(2,3). Inaccurate measurements may lead to suboptimal refractive outcomes and an increased risk of postoperative complications(4).
Placido-based corneal topographers, such as the OPD-Scan III (Nidek, Japan), are widely used to assess anterior corneal curvature and perform aberrometry(5,6). Although these devices provide robust anterior surface evaluation, they are inherently limited in their ability to detect posterior corneal changes, which are critical for the early identification of corneal ectasia(7). In contrast, Scheimpflug-based tomographers acquire cross-sectional images of the cornea and reconstruct three-dimensional maps, enabling simultaneous analysis of anterior and posterior corneal surfaces as well as detailed pachymetric profiles(8-10).
Pentacam (Oculus, Germany) is the most extensively validated Scheimpflug device and is often considered the clinical reference standard for corneal tomography(11). However, its high cost limits accessibility in resource-constrained settings(12). Scansys (Mediworks, China) is a newer Scheimpflug-based tomographer designed as a more affordable alternative. It captures up to 60 images per scan and incorporates artificial intelligence–based algorithms to enhance data processing and measurement reproducibility(13). Preliminary studies have demonstrated strong agreement with established Scheimpflug systems, such as Sirius, suggesting potential clinical reliability(14-16). Nevertheless, comparative validation against devices based on different optical principles, particularly Placido-based topographers, remains limited.
Given the clinical importance of accurate corneal imaging for refractive and cataract surgery planning and the potential impact of cost-effective diagnostic technologies, a rigorous evaluation of Scansys is warranted. Therefore, this study aimed to assess the reliability and comparability of corneal measurements obtained with Scansys in healthy eyes, using the OPD-Scan III and specular microscopy as reference devices.
METHODS
Study design and ethical approval
This cross-sectional study with prospective data collection was conducted at Hospital dos Olhos, Bragança Paulista, São Paulo, Brazil, between January and December 2023. The study protocol was approved by the Institutional Review Board of Irmandade Santa Casa de Misericórdia de Santos (CAAE: 89803324.7.0000.0139) and adhered to the tenets of the Declaration of Helsinki. All participants provided written informed consent after receiving a detailed explanation of the study procedures and potential risks.
Study population
Forty consecutive patients (80 eyes) were recruited from the cornea outpatient clinic. Eligible participants were aged 18–95 yr and had no history of keratorefractive surgery, corneal dystrophies, corneal scars, or ocular diseases such as glaucoma or uveitis. Contact lens users were instructed to discontinue lens wear for at least 7 days before examination to minimize corneal warpage. Eyes with poor fixation, severe dry eye, or any condition preventing reliable image acquisition were excluded.
Examination protocol
All participants underwent corneal imaging using three devices: Scansys (Mediworks, China), OPD-Scan III (Nidek, Japan), and specular microscopy (Tomey EM-4000, Tomey Corporation, Japan). The order of device use was randomized using computer-generated sequences. All measurements were performed by a single experienced operator under standardized environmental conditions (temperature: 22°C–24°C; humidity: 40%–60%). Examinations were conducted in the same room under standardized ambient lighting; participants remained in the room for 2 min for adaptation before image acquisition, although ambient illumination was not objectively quantified. Patients were positioned using chin and forehead rests and instructed to fixate on the internal target of each device. All devices were operated according to the manufacturers' instructions without additional study-specific calibration.
Measurement protocol and quality control
Each eye was measured three times with each device. For interdevice agreement analyses, the highest-quality acquisition per eye/device was selected from the three scans. Acquisitions flagged as unreliable were repeated, up to five attempts. If acceptable quality was not achieved, the corresponding eye/parameter was excluded. For intradevice repeatability analyses, all three acquisitions were included. For specular microscopy, the mean pachymetry value provided by the device was used. Because specular microscopy measurements were intended for descriptive context only, no formal interdevice agreement analyses were performed for this device.
Device description
Scansys employs Scheimpflug-based tomography, capturing up to 60 cross-sectional images per scan and reconstructing three-dimensional corneal maps, pachymetric profiles, and wavefront data. The OPD-Scan III uses Placido-disk technology to evaluate anterior corneal curvature and aberrometry. Specular microscopy was used to assess endothelial parameters and central corneal thickness.
Parameters evaluated
Analyzed parameters included flat keratometry (K1), steep keratometry (K2), keratometric axis, corneal astigmatism magnitude, asphericity (Q-value), photopic pupil diameter, horizontal and vertical kappa distance, corneal diameter (white-to-white), corneal refractive power, higher-order aberrations, and central corneal thickness.
Outcomes
The primary outcomes were agreement and comparability of K1 and K2 between Scansys and OPD-Scan III. Secondary outcomes included corneal asphericity, photopic pupil diameter, and kappa-related metrics. Specular microscopy measurements were reported descriptively.
Sample size
An a priori sample size was estimated using G*Power for paired-sample comparisons. Assuming a conservative small-to-moderate within-eye standardized effect size (dz=0.40), a two-sided α of 0.05, and 80% power, the required number of paired eyes was 52. We planned to enroll 80 eyes (40 participants) to account for within-subject correlation due to the inclusion of both eyes and for anticipated exclusions related to image quality and incomplete paired measurements at the eye or parameter level.
Statistical analysis
Statistical analyses were performed using SPSS version 26.0 (IBM Corp., Armonk, New York) and Minitab version 21.2 (Minitab Inc., State College, Pennsylvania). Because measurements from both eyes of some participants were included, intereye correlation was addressed using generalized estimating equations (GEE) with participant as the clustering unit and robust (sandwich) standard errors. Descriptive statistics included means, standard deviations, coefficients of variation, and 95% confidence intervals. Data normality was assessed using the Kolmogorov–Smirnov test.
Between-device comparisons were conducted using GEE models with participant-level clustering. When paired measurements from the same eye were available, within-eye differences were estimated accordingly. Independent-sample comparisons were presented for completeness but were not considered the primary inferential approach. Agreement between devices was assessed using Bland–Altman analysis, including mean differences and 95% limits of agreement. Intraclass correlation coefficients (ICC) were calculated to evaluate intradevice repeatability, with values >0.75 considered indicative of good reproducibility. Pearson correlation coefficients were used to assess associations between continuous variables, and outliers were identified using Tukey's method.
Although a sample size of 40 patients was planned, exclusions at the eye or parameter level resulted in variable effective sample sizes across outcomes, potentially reducing precision for some estimates. Because K1 and K2 were prespecified as primary outcomes, all other comparisons were considered secondary; no adjustment for multiple comparisons was applied, and secondary findings were interpreted accordingly. A p-value <0.05 was considered statistically significant.
RESULTS
A total of 80 eyes from 40 patients (mean age: 42.3 ± 15.7 yr; 55% female) were included. All participants completed the study protocol, and no adverse events were reported. After quality control, 59–67 eyes per parameter were available for independent-sample analyses and 48–51 eyes for paired-eye analyses. Exclusions occurred only when acceptable image quality could not be obtained, resulting in variable denominators across outcomes.
Although the cohort primarily comprised healthy eyes, one to three eyes showed topographic or tomographic findings suggestive of keratoconus (Kmax >47.2 D or thinnest pachymetry <470 μm). These cases were insufficient for subgroup analysis and were excluded from comparative evaluations.
Comparative analysis using independent samples
Table 1 summarizes the independent-sample comparison between Scansys and OPD-Scan III. No statistically significant differences were observed in corneal astigmatism magnitude, keratometric axes, or higher-order aberrations. However, significant interdevice differences were detected for Q-value (p=0.024), photopic pupil diameter (p<0.001), and kappa distance (p<0.001). Scansys consistently yielded more negative asphericity values, smaller photopic pupil diameters, and shorter kappa distances compared with OPD-Scan III. These independent-sample analyses are presented descriptively; inferential interpretation was prioritized in paired-eye analyses when valid measurements from both devices were available for the same eye.
Paired-eye agreement and repeatability
Paired-eye analyses (Table 2) corroborated these findings. Significant differences between devices persisted for Q-value, photopic pupil diameter, and kappa distance. Additionally, small but statistically significant differences were observed for flat and steep keratometry (K1: p=0.003; K2: p=0.002), with Scansys consistently reporting slightly higher values. Although the mean paired-eye differences were modest (approximately 0.17–0.18 D), such discrepancies may be clinically relevant for intraocular lens power calculations and centration of premium IOLs. Mean absolute differences were approximately 0.30–0.32 D, indicating that individual-level disagreement may exceed the average systematic bias. Astigmatism magnitude and keratometric axes remained comparable between devices. All inferential comparisons accounted for within-subject correlation through participant-level clustering.
Absolute differences and variability
Absolute differences and coefficients of variation are presented in Table 3. Variability was particularly pronounced for steep keratometry (mean absolute difference: 0.30 D; CV: 106%) and photopic pupil diameter (mean absolute difference: 0.67 mm; CV: 103%). Angular parameters, such as kappa orientation, demonstrated the greatest dispersion (CV: 195%), indicating substantial interdevice variability at the individual-level despite relatively consistent group means. These findings further support the limited interchangeability of the two devices without calibration.
Reproducibility and pachymetric findings
Scansys demonstrated high intradevice reproducibility, with ICC values exceeding 0.75 for K1, K2, Q-value, and central corneal thickness (CCT). Specular microscopy pachymetry was included for descriptive purposes only, using the device-provided mean value. The thinnest corneal thickness measured by specular microscopy was 533.0 ± 9.6 μm (CV: 8%). No formal agreement analyses were conducted between specular microscopy and the other devices; therefore, these results should be interpreted as exploratory.
Graphical analysis
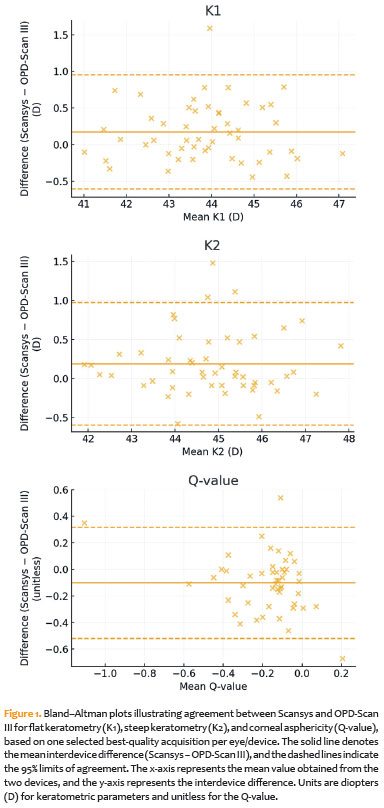
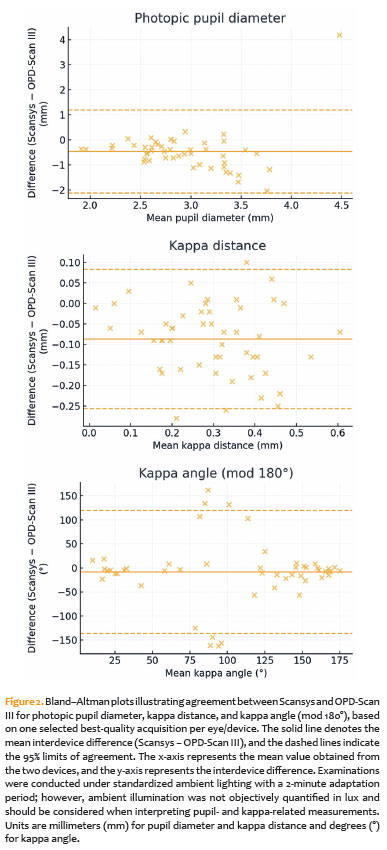
Bland–Altman plots for keratometric parameters are shown in Figure 1, illustrating bias and 95% limits of agreement between Scansys and OPD-Scan III for Q-value, K1, and K2. Differences in photopic pupil diameter, kappa distance, and kappa angle are presented in Figure 2, highlighting device-specific measurement tendencies, particularly under varying illumination conditions. Because ambient illumination was standardized but not objectively quantified in lux, pupil- and kappa-related findings should be interpreted with this limitation in mind.
DISCUSSION
This study evaluated the reliability and comparability of corneal measurements obtained with a Scheimpflug-based tomographer (Scansys) relative to a Placido-based topographer (OPD-Scan III) and specular microscopy in healthy eyes. The principal findings indicate that Scansys demonstrated high intradevice reproducibility for keratometry, Q-value, and CCT. However, statistically significant interdevice differences were observed for flat and steep keratometry, corneal asphericity, pupil diameter, and kappa distance. Absolute variability was substantial across several parameters—particularly angular metrics—highlighting that these devices should not be used interchangeably. Beyond mean bias, considerable individual-level dispersion was evident for several measurements, emphasizing that interchangeability must be evaluated on a parameter-specific basis rather than inferred from group averages alone.
Differences in keratometry (K1 and K2) were statistically significant, with Scansys yielding slightly higher values in paired-eye comparisons (approximately 0.17–0.18 D). These results are consistent with previous studies comparing Placido- and Scheimpflug-based systems, which have reported systematic discrepancies attributable to fundamental differences in measurement principles. Placido-based devices rely exclusively on anterior surface reflections, whereas Scheimpflug tomography incorporates information from both anterior and posterior corneal surfaces, often resulting in marginally higher keratometric values(5,8,11). Differences exceeding 0.25 D have been shown to influence IOL power calculations, particularly in eyes with irregular corneas or a history of refractive surgery(17). Thus, even modest interdevice discrepancies may have clinically meaningful consequences. Moreover, the mean absolute differences observed in the present study (approximately 0.30–0.32 D) indicate that disagreement at the individual-eye level may exceed the average systematic bias, further supporting the recommendation to avoid cross-device substitution when keratometry is used for IOL power calculation or longitudinal monitoring. Clinically, a systematic offset of approximately 0.17–0.18 D—combined with absolute differences approaching 0.30 D—may affect refractive predictability in individual cases, particularly when targeting emmetropia, selecting premium IOLs, or operating near clinical decision thresholds.
The more negative Q-values measured with Scansys are consistent with prior reports showing that Scheimpflug-based devices more effectively capture peripheral corneal flattening and posterior elevation(18,19). Accurate evaluation of corneal asphericity is essential for early detection of keratoconus and for planning customized refractive ablation profiles. Previous evidence suggests that Scheimpflug-derived Q-values may be more sensitive in differentiating subclinical ectasia from normal corneas(20). Similarly, Scansys consistently measured smaller photopic pupil diameters than the OPD-Scan III. This discrepancy is likely related to differences in device-specific illumination protocols, a phenomenon previously documented, with reported differences of up to 0.5 mm between Pentacam and OPD-Scan III under varying lighting conditions(21). Because pupil size directly affects refractive surgery outcomes and the performance of multifocal IOLs, these measurements should be interpreted cautiously. In the present study, ambient illumination was standardized with a fixed adaptation period but was not objectively quantified in lux; therefore, pupil- and kappa-related differences should be considered in light of potential residual illumination effects and device-specific internal lighting geometry.
Kappa distance was consistently shorter when measured with Scansys, likely reflecting differences in reference alignment, as Placido-based systems typically reference the pupil center, whereas Scheimpflug devices align to the corneal vertex. This methodological distinction may account for the observed discrepancies and is clinically relevant, as decentration exceeding 0.3 mm has been associated with reduced optical quality in eyes implanted with diffractive trifocal IOLs(22). Although mean differences were systematic, variability in the kappa angle was high, underscoring the challenges of achieving cross-device agreement for alignment-related parameters. These findings suggest that direct substitution of alignment metrics across platforms should be avoided, particularly in surgical planning and longitudinal follow-up, where consistency of measurement reference is critical.
With respect to pachymetry, descriptive agreement was observed between Scansys and specular microscopy. The mean thinnest corneal thickness measured by specular microscopy (533.0 ± 9.6 μm) was consistent with values obtained using Scansys. Previous studies comparing Scheimpflug tomography with specular and ultrasound pachymetry have generally reported good agreement(23-30). Although the present study did not include formal statistical agreement analyses for this comparison, the descriptive concordance suggests that Scansys may provide clinically reliable pachymetric measurements. Specular microscopy pachymetry was intentionally included for descriptive context only, as the study was not designed or powered to support a dedicated intermethod agreement analysis. Consequently, no Bland–Altman or correlation analyses were performed, and these findings should be interpreted as exploratory.
Recent literature further supports the conclusion that agreement is highly parameter-dependent, even among tomographers based on similar optical principles. Khorrami-Nejad et al. reported partial interchangeability between Scansys and Sirius for selected anterior segment parameters while cautioning against broad substitution(31). Similarly, Hashemian et al.(32) found parameter-specific agreement when comparing Scansys with Pentacam HR, emphasizing that interchangeability should not be generalized across outputs. Xu et al.(33) also reported good repeatability and agreement between a reference Scheimpflug tomographer and a low-cost Scheimpflug system for selected parameters, while discouraging claims of universal interchangeability. Collectively, these findings are consistent with the present results and reinforce the need for metric-specific interpretation when considering cross-device use.
Several limitations should be acknowledged. First, although the cohort was representative of routine clinical practice, only one to three eyes exhibited features suggestive of keratoconus, precluding meaningful subgroup analyses in ectatic corneas and limiting generalizability to diseased eyes. Second, Pentacam—the most widely validated Scheimpflug tomographer—was not available during the study period; therefore, Scansys could not be directly compared with the current clinical reference standard. Third, pachymetric comparisons with specular microscopy were descriptive rather than statistical, limiting conclusions regarding formal agreement. Fourth, all measurements were obtained by a single operator, which minimized interoperator variability but may have introduced systematic bias. Additionally, quality-control exclusions and parameter-specific acquisition failures resulted in variable denominators across analyses, potentially reducing precision for some outcomes. Ambient illumination was not quantified in lux, which is a relevant limitation for pupil- and kappa-related parameters, and intersession repeatability was not assessed.
Despite these limitations, the findings have practical clinical implications. Scansys demonstrated high intradevice reproducibility and generally acceptable agreement with OPD-Scan III and specular microscopy for most parameters. Its affordability and incorporation of artificial intelligence–based processing may facilitate broader access to reliable corneal imaging, particularly in resource-limited settings. Nevertheless, given the observed interdevice variability, consistent use of the same device is recommended for preoperative assessment and longitudinal follow-up. This recommendation is especially critical for keratometry, corneal asphericity, and pupil- and alignment-related metrics, where systematic offsets and increased dispersion may confound clinical interpretation if devices are alternated.
In conclusion, Scansys provides reproducible and reliable corneal measurements in healthy eyes; however, systematic differences relative to Placido-based devices indicate that it should not be used interchangeably with the OPD-Scan III. Although its cost-effectiveness makes it a promising option for routine anterior segment evaluation, further studies—including investigations in ectatic corneas and direct comparisons with Pentacam—are needed to better define its clinical role. Future research should also incorporate objective illumination control, formal agreement analyses for pachymetry when used as a comparative endpoint, and assessments of intersession and interoperator repeatability to establish parameter-specific thresholds for clinical interchangeability.
AUTHORS' CONTRIBUTIONS:
Significant contribution to conception and design: Camyla Lemos Budib, Caio Henrique Peres Oliani, Guilherme de Oliveira Ferreira, Ulysses Tachibana, Jose Alvaro Pereira Gomes, Bernardo Kaplan Moscovici, Marcello Novoa Colombo-Barboza, Guilherme Novoa Colombo-Barboza. Data Acquisition: Camyla Lemos Budib, Caio Henrique Peres Oliani, Guilherme de Oliveira Ferreira, Ulysses Tachibana, Bernardo Kaplan Moscovici, Marcello Novoa Colombo-Barboza, Guilherme Novoa Colombo-Barboza. Data Analysis and interpretation: Camyla Lemos Budib, Caio Henrique Peres Oliani, Guilherme de Oliveira Ferreira, Ulysses Tachibana, Jose Alvaro Pereira Gomes, Bernardo Kaplan Moscovici, Marcello Novoa Colombo-Barboza, Guilherme Novoa Colombo-Barboza. Manuscript Drafting: Camyla Lemos Budib, Caio Henrique Peres Oliani, Guilherme de Oliveira Ferreira, Ulysses Tachibana, Bernardo Kaplan Moscovici, Marcello Novoa Colombo-Barboza, Guilherme Novoa Colombo-Barboza. Significant intellectual contente revision of the manuscript: Jose Alvaro Pereira Gomes, Bernardo Kaplan Moscovici, Marcello Novoa Colombo-Barboza, Guilherme Novoa Colombo-Barboza. Final approval of the submitted manuscript: Camyla Lemos Budib, Caio Henrique Peres Oliani, Guilherme de Oliveira Ferreira, Ulysses Tachibana, Jose Alvaro Pereira Gomes, Bernardo Kaplan Moscovici, Marcello Novoa Colombo-Barboza, Guilherme Novoa Colombo-Barboza. Statistical analysis: Bernardo Kaplan Moscovici. Obtaining funding: not applicable. Supervision of Administrative, technical, or material support: Marcello Novoa Colombo-Barboza. Research group leadership: Bernardo Kaplan Moscovici, Marcello Novoa Colombo-Barboza, Guilherme Novoa Colombo-Barboza.
REFERENCES
1. Fan R, Chan TC, Prakash G, Jhanji V. Applications of corneal topography and tomography: a review. Clin Exp Ophthalmol. 2018;46(2):133–46.
2. Daxecker F. Christoph Scheine's eye studies. Doc Ophthalmol. 1992;81(1):27–35.
3. Seitz B, Behrens A, Langenbucher A. Corneal topography. Curr Opin Ophthalmol. 1997;8(4):8–24.
4. Douthwaite WA. EyeSys corneal topography measurement applied to calibrated ellipsoidal convex surfaces. Br J Ophthalmol. 1995;79(9):797–801.
5. Motlagh MN, Moshirfar M, Murri MS, Skanchy DF, Momeni-Moghaddam H, Ronquillo YC, et al. Pentacam® corneal tomography for screening of refractive surgery candidates: a review of the literature, Part I. Med Hypothesis Discov Innov Ophthalmol. 2019;8(3):177–203.
6. Koch DD, Foulks GN, Moran CT, Wakil JS. The Corneal EyeSys System: accuracy analysis and reproducibility of first-generation prototype. Refract Corneal Surg. 1989;5(6):424–9.
7. Müller-Breitenkamp U, Hockwin O. Scheimpflug photography in clinical ophthalmology. A review. Ophthalmic Res. 1992;24(Suppl 1):47–54.
8. Nilforoushan MR, Speaker M, Marmor M, Abramson J, Tullo W, Morschauser D, et al. Comparative evaluation of refractive surgery candidates with Placido topography, Orbscan II, Pentacam, and wavefront analysis. J Cataract Refract Surg. 2008;34(4):623–31.
9. Cao K, Verspoor K, Sahebjada S, Baird PN. Evaluating the performance of various machine learning algorithms to detect subclinical keratoconus. Transl Vis Sci Technol. 2020;9(2):24.
10. Zhang X, Munir SZ, Sami Karim SA, Munir WM. A review of imaging modalities for detecting early keratoconus. Eye (Lond). 2021;35(1):173–87.
11. Uçakhan ÖÖ, Cetinkor V, Özkan M, Kanpolat A. Evaluation of Scheimpflug imaging parameters in subclinical keratoconus, keratoconus, and normal eyes. J Cataract Refract Surg. 2011;37(6):1116–24.
12. Huo Y, Xie R, Chen X, Li S, Zou H, Liu Y, et al. Comparison of a Scheimpflug imaging with other screening indices in diagnosing keratoconus and keratoconus suspect. Sci Rep. 2024;14(1):23187.
13. Hashemi H, Beiranvand A, Yekta A, Maleki A, Yazdani N, Khabazkhoob M. Pentacam top indices for diagnosing subclinical and definite keratoconus. J Curr Ophthalmol. 2016;28(1):21–6.
14. Dong S, Xu R, Wang Y, Xiao W, Jiao Y. Comparisons of biometric parameters measurements by OPD-SCAN Ⅲ and Pentacam in cataract. Photodiagnosis Photodyn Ther. 2024;45:103911.
15. Asgari S, Hashemi H, Jafarzadehpur E, Mohamadi A, Rezvan F, Fotouhi A. OPD-Scan III: a repeatability and inter-device agreement study of a multifunctional device in emmetropia, ametropia, and keratoconus. Int Ophthalmol. 2016;36(5):697–705.
16. Nguyen N, Gorovoy M. Specular microscopy and confocal microscopy. Curr Opin Ophthalmol. 2018;29(4):333–9.
17. Bao T, Yin L, Liu C, Pang Y, Zhang G, Yang Y, et al. Agreement of anterior segment measurements between LenStar LS 900 optical biometer and OPD Scan III wavefront aberrometer devices in eyes with cataract. Photodiagnosis Photodyn Ther. 2023;41:103207
18. Wan KH, Liao XL, Yu M, Tsui RW, Chow VW, Chong KK, et al. Wavefront aberrometry repeatability and agreement-A comparison between Pentacam AXL Wave, iTrace and OPD-Scan III. Ophthalmic Physiol Opt. 2022;42(6):1326–37.
19. Belknap SM, Georgopoulos R, West DP, et al. Clinical reference guide to corneal topography devices. J Cataract Refract Surg. 2017;43(8):1103–10.
20. Rabinowitz YS. Videokeratographic indices to aid in screening for keratoconus. J Refract Surg. 1995;11(5):371–9.
21. Savini G, Barboni P, Carbonelli M, Hoffer KJ. Repeatability of automatic measurements by a new Scheimpflug camera combined with Placido topography. J Cataract Refract Surg. 2011;37(10):1809–16.
22. Nusair O, Asadigandomani H, Farrokhpour H, Moosaie F, Bibak-Bejandi Z, Razavi A, Daneshvar K, Soleimani M. Clinical Applications of Artificial Intelligence in Corneal Diseases. Vision (Basel). 2025 Aug 18;9(3):71.
23. Chen D, Lam AK. Intrasession and intersession repeatability of the Pentacam system on posterior corneal assessment in the normal human eye. J Cataract Refract Surg. 2007;33(3):448–54.
24. Rodrigues PF, Moscovici BK, Lamazales L, Freitas MM, Gomes JÁ, Nosé W, et al. Measurement of the visual axis through two different methods: quantification and differences for measuring chord μ. Arq Bras Oftalmol. 2023;87(4):e2022–0035.
25. Rios LC, Silva PG, Canamary Junior AM, Rodrigues P, Regatieri CV, Campos M, et al. Centration in refractive surgery. Arq Bras Oftalmol. 2020;83(1):76–81.
26. Carvalho KA, Oliveira NA, Melice RS, Rocha RH, Rodrigues PF, et al. Comparison of Pentacam data in patients with diagnostic criteria for keratoconus in only one eye. Rev Bras Oftalmol. 2024;83:e0047.
27. Gouvea L, Alshaker S, Rocha KM, Chamon W, Chan CC, Rootman DS. Effect of monofocal, wavefront-shaped, and diffractive trifocal intraocular lenses on scanning-slit automated refraction. Am J Ophthalmol. 2025;269:450–6.
28. Kamiya K, Iijima K, Nobuyuki S, Mori Y, Miyata K, Yamaguchi T, Shimazaki J, Watanabe S, Maeda N. Predictability of Intraocular Lens Power Calculation for Cataract with Keratoconus: A Multicenter Study. Sci Rep. 2018 Jan 22;8(1):1312.
29. Ghoreishi SM, Mortazavi SAA, Abtahi ZA, Abtahi MA, Sonbolestan SA, Abtahi SH, Mohammadinia M, Isfahani KN. Comparison of Scheimpflug and swept-source anterior segment optical coherence tomography in normal and keratoconus eyes. Int Ophthalmol. 2017 Aug;37(4):965-971.
30. Vinciguerra P, Albè E, Trazza S, Rosetta P, Vinciguerra R, Seiler T, et al. Refractive, topographic, tomographic, and aberrometric analysis of keratoconic eyes undergoing corneal cross-linking. Ophthalmology. 2009;116(3):369–78.
31. Khorrami-Nejad M, Khodaparast M, Abdulkadhim IA, Azizi E, Rashidi F, Damanpak V, et al. A comparison of Scansys and Sirius tomography in healthy eyes. BMC Ophthalmol. 2024;24(1):138.
32. Hashemian H, Khodaparast M, Khorrami-Nejad M, Mohmmed MH, Ahmadzadeh H, Hamedani MA, et al. Comparison of Scansys and Pentacam HR in healthy eyes. Expert Rev Ophthalmol. 2024;19(6):467–74.
33. Xu W, Zhai C, Yusufu M, Gan J, Li S, Zheng Y, et al. Repeatability and agreement between a reference Scheimpflug tomographer and a low-cost Scheimpflug system. J Cataract Refract Surg. 2023;49(6):614–9.
Data Availability Statement: The datasets produced and/or analyzed in this study can be provided to referees upon request.
Edited by
Editor-in-Chief: Newton Kara-Júnior
Associate Editor: Dácio C. Costa
Submitted for publication:
September 5, 2025.
Accepted for publication:
January 23, 2026.
Approved by the following research ethics committee: Irmandade Santa Casa de Misericórdia de Santos – ISCMS (CAAE: 89803324.7.0000.0139).
Funding: This study received no specific financial support.
Disclosure of potential conflicts of interest: The authors declare no potential conflicts of interest.
How to cite this article: