Arq. Bras. Oftalmol. 2026; 89 (2): 10.5935/0004-2749.2025-0257
Total: 528
Ali Osman Gündogan1; Refik Oltulu1; Selman Belviranli1; Ali Tezcan2; Selami Aykut Temiz3; Esra Altay3
DOI: 10.5935/0004-2749.2025-0257
ABSTRACT
PURPOSE: This study aimed to analyze ocular surface parameters and evaluate meibomian gland dysfunction using meibography in newly diagnosed patients with atopic dermatitis compared with healthy controls.
METHODS: This cross-sectional clinical study included 51 newly diagnosed patients with atopic dermatitis and 45 age- and sex-matched healthy controls. Disease severity was assessed using the Eczema Area Severity Index. The Ocular Surface Disease Index questionnaire, Schirmer test, tear meniscus height, noninvasive tear break-up time, conjunctival redness grading, and meibography staging were performed. Meibomian gland dropout was graded for each eyelid from 0 (no loss) to 3 (loss of >2/3 of the total gland area).
RESULTS: Schirmer test values, tear meniscus height, and noninvasive tear break-up time were significantly lower in the atopic dermatitis group than in the control group (p<0.001, p<0.001, and p<0.001, respectively). In contrast, Ocular Surface Disease Index scores, conjunctival redness grades, and total meiboscores were significantly higher in the atopic dermatitis group than in the control group (p<0.001, p<0.001, and p<0.001, respectively). Moreover, in the atopic dermatitis group, a significant positive correlation was observed between the Eczema Area Severity Index score and total meiboscore (rₛ=0.390, p=0.005), while a significant negative correlation was found between the Eczema Area Severity Index score and Schirmer test results (rₛ=−0.301, p=0.032). Conclusions: Newly diagnosed patients with atopic dermatitis exhibit significant alterations in tear film parameters and meibomian gland morphology compared with healthy individuals. These patients should be monitored for early development of dry eye disease and meibomian gland dysfunction to prevent associated complications.
Keywords: Dermatitis, atopic; Ocular surface; Meibomian gland dysfunction; Meibography; Dry eye syndromes
INTRODUCTION
Atopic dermatitis (AD) is a chronic inflammatory skin disease characterized by recurrent, pruritic eczematous lesions. It can occur at any age and affects approximately 20%-25% of children and 2%-3% of adults(1). Increasing evidence suggests that AD is associated with extracutaneous manifestations, among which ocular involvement is common. The pathogenesis of ocular complications in AD is complex and multifactorial, involving immune dysregulation, mechanical trauma from eye rubbing, medication-related effects, and genetic predisposition(2).
Symptoms such as itching, tearing, and stinging, frequently reported by patients with AD, are often linked to allergic ocular involvement. Chronic corneal irritation in an atopic environment may adversely affect corneal integrity and predispose patients to ectatic disorders(3,4). Several ocular pathologies, including cataract, retinal detachment, blepharitis, glaucoma, keratoconjunctivitis, and keratoconus, have been reported in association with AD(5-7).
Meibomian glands (MGs) are specialized sebaceous glands located within the eyelids, with 20-30 glands in the lower eyelid and 30-40 glands in the upper eyelid. These glands secrete lipids essential for maintaining tear film stability and preventing excessive evaporation(8). Meibomian gland dysfunction (MGD) is a chronic disorder characterized by terminal duct obstruction and/or qualitative or quantitative alterations in glandular secretion(9). MGD is the leading cause of dry eye disease (DED), accounting for nearly two-thirds of cases(10).
MGD and dry eye have been reported in association with atopic keratoconjunctivitis (AKC), a severe ocular complication of AD. Reduced tear break-up time, goblet cell loss, and conjunctival squamous metaplasia have been documented in these patients and are thought to contribute to tear film instability(11). However, to date, MGD in patients with AD has not been systematically evaluated using meibography.
Therefore, this study aimed to assess ocular surface parameters and evaluate meibomian gland morphology using meibography in newly diagnosed patients with AD.
METHODS
Study population and design
This cross-sectional clinical study included patients newly diagnosed with AD who were referred from the Dermatology Clinic of Necmettin Erbakan University Faculty of Medicine Hospital between September 2024 and December 2024. The study group consisted of the right eyes of 51 patients with AD. Disease severity was assessed using the globally accepted Eczema Area Severity Index (EASI), which evaluates four anatomical regions based on the extent and severity of erythema, induration/papulation, excoriation, and lichenification, yielding a total score ranging from 0 to 72(1).
Exclusion criteria
The exclusion criteria were as follows:
1. Systemic diseases affecting the ocular surface (e.g., connective tissue diseases, autoimmune disorders, diabetes mellitus, and dyslipidemia).
2. Ocular surface or eyelid disorders (e.g., conjunctivitis, keratitis, and eyelid abnormalities).
3. Contact lens use within the previous 6 months.
4. History of intraocular or periocular surgery.
5. Smoking or alcohol consumption.
6. Use of topical ocular medications.
7. Use of systemic medications affecting the ocular surface or meibomian glands.
The control group consisted of the right eyes of 42 age- and sex-matched healthy individuals presenting for routine ophthalmic examination. The study adhered to the Declaration of Helsinki and was approved by the Necmettin Erbakan University Ethics Committee (No. 2024/5152). Written informed consent was obtained from all participants.
Examination protocol and measurements
All participants underwent a comprehensive ophthalmologic examination, including best-corrected visual acuity measurement, biomicroscopic anterior segment examination, fundus examination, and intraocular pressure measurement. Subsequently, the Ocular Surface Disease Index (OSDI) questionnaire, Schirmer test, tear meniscus height (TMH), noninvasive tear break-up time (NITBUT), meibography staging, and conjunctival redness assessments were performed. All noninvasive tests were conducted prior to the Schirmer test.
The examination sequence was as follows. First, the OSDI questionnaire was administered. It was followed by keratographic measurements, including NITBUT, TMH, conjunctival redness assessment, and meibography. After the completion of these procedures, the Schirmer test was performed without topical anesthesia.
The OSDI questionnaire consists of three sections containing five, four, and three questions. Ocular symptoms were scored from 0 to 4 based on frequency (0: never, 1: sometimes, 2: half of the day, 3: most of the time, 4: always). The total OSDI score ranges from 0 to 100 and was calculated using the following formula: total score × 25 / number of valid questions.
For the Schirmer test, a 5 × 35 mm filter paper strip was placed in the conjunctival fornix at the lateral one-third of the lower eyelid without topical anesthesia. After 5 minutes, the length of wetting was measured in millimeters. Values <10 mm were considered abnormal. Tear break-up time (TBUT) and Schirmer tests were performed by the same ophthalmologist (AOG).
The Keratograph 5M (Oculus; Wetzlar, Germany) was used to measure TMH, NITBUT, meibography stage, and conjunctival redness. All procedures were performed by the same ophthalmologist (AT) under standardized mesopic lighting and room conditions, and measurements were obtained at the same time of day. Three measurements were obtained from each eye, and the mean values were used for analysis.
NITBUT was defined as the time between the last complete blink and the decay of the Placido ring pattern reflected on the tear film surface. The Keratograph 5M provides two NITBUT parameters: NITBUT-first, defined as the time to the first tear film disruption, and NITBUT-average, defined as the mean time of all tear film break-up events within an 8-mm corneal diameter(12).
TMH was measured after disabling the red concentric ring illumination and using a dark background. Images were captured at three locations along the lower eyelid—central, temporal limbus, and nasal limbus—while participants were imaged in the primary gaze position. Following three blinks over 3 seconds, the vertical distance between the eyelid margin and the tear meniscus was measured along imaginary vertical lines passing through the pupil center, temporal limbus, and nasal limbus.
Conjunctival redness was assessed using the Keratograph 5M and analyzed by the device’s software, which calculates scores based on the percentage ratio of vascularized areas to the total analyzed area(13). For example, a ratio of 10% corresponds to a score of 1.0. Scores range from 0.0 to 4.0, with a maximum ratio of 40%, and were recorded for each participant.
MGs were imaged using the infrared camera system of the Keratograph 5M. MG loss was evaluated subjectively using a grading scale from 0 to 3 (grade 0: no gland loss; grade 1: <33% loss; grade 2: 33%-67% loss; grade 3: >67% loss)(14). Meiboscore assessments were performed by the same investigator (AT). Upper and lower eyelids were evaluated separately, and total meiboscores were calculated by adding the upper and lower eyelid scores for each eye.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics version 29.0 (IBM Corp., Armonk, NY, USA). Continuous variables were expressed as mean ± standard deviation (SD), while categorical variables were expressed as counts (n) and percentages (%). Normality of continuous variables was assessed using the Kolmogorov-Smirnov test. The Mann-Whitney U test and Pearson’s chi-square test were used to compare continuous and categorical variables, respectively. A two-sided p-value of <0.05 was considered statistically significant.
RESULTS
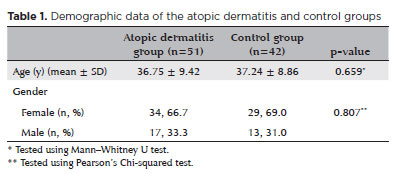
Of the 51 newly diagnosed patients with AD included in the study, 17 (33.3%) and 34 (66.7%) were males and females, respectively. In the control group (n=42), 13 (31%) and 29 (69%) were males and females, respectively. The mean age was 36.75 ± 9.42 and 37.24 ± 8.86 years in the AD and control groups, respectively. No significant differences were observed between the groups with respect to age or sex distribution (p=0.659 and p=0.807, respectively; Table 1).
The mean Schirmer test value was significantly lower in the AD group (7.88 ± 3.02 mm) than in the control group (11.74 ± 2.37 mm, p<0.001). NITBUT-first was 5.65 ± 2.41 and 9.37 ± 1.44 s, and NITBUT-average was 9.88 ± 3.07 and 15.56 ± 3.27 s in the AD and control groups, respectively. Both NITBUT-first and NITBUT-average values were significantly lower in the AD group than in the control group (p<0.001).
Temporal TMH was 0.20 ± 0.05 and 0.26 ± 0.03 mm, and central TMH was 0.17 ± 0.04 and 0.23 ± 0.02 mm, and nasal TMH was 0.21 ± 0.05 and 0.26 ± 0.03 mm, in the AD and control groups, respectively. All TMH measurements were significantly lower in the AD group than in the control group (p<0.001).
The mean OSDI score was significantly higher in the AD group (35.44 ± 7.44) than in the control group (14.91 ± 5.40, p<0.001). Similarly, conjunctival redness scores were significantly higher in the AD group (1.42 ± 0.33) than in the control group (1.15 ± 0.33, p<0.001). The mean total meiboscore (upper + lower eyelids) was also significantly higher in the AD group (1.70 ± 1.56) than in the control group (0.55 ± 0.50, p<0.001; Table 2).
Correlation analyses within the AD group demonstrated a significant positive correlation between the EASI score and total meiboscore (rs=0.390, p=0.005), as well as a significant negative correlation between EASI score and Schirmer test values (rs=−0.301, p=0.032; Table 3).
Representative images of TMH, NITBUT, conjunctival redness, and meibography in patients with AD are shown in figure 1, and corresponding images from the control group are shown in figure 2.
DISCUSSION
Our investigation demonstrated increased conjunctival redness and MG loss in patients with newly diagnosed AD. In addition, a positive correlation was observed between disease severity, as assessed by EASI and the degree of MG loss. Significant reductions in TMH, NITBUT, and Schirmer test scores were also detected in patients with AD. Conversely, a negative correlation was identified between EASI scores and Schirmer test results.
AD is a common inflammatory skin disorder that typically begins in early childhood and may persist into adulthood(15). Because the ocular surface can be affected by systemic inflammation associated with AD, ocular or eyelid involvement has been reported in 20%-43% of patients(16). Recent studies have emphasized that ocular surface disorders, including conjunctivitis, keratitis, keratoconus, keratoconjunctivitis, DED, and blepharitis, occur more frequently in patients with AD(17).
AKC is a bilateral, chronic inflammatory condition of the conjunctiva and eyelids that is closely associated with AD. Although the precise mechanisms underlying allergic inflammation in AKC remain incompletely understood, T-cell-mediated immune responses and infiltration by mast cells, eosinophils, and other inflammatory cells are known to play major roles. Histologically, skin inflammation in AD is characterized by dense infiltration of lymphocytes, monocytes, and eosinophils, which secrete proinflammatory cytokines and reactive oxygen species in response to immunologic and nonimmunologic stimuli(18,19). Wakamatsu et al. reported that ocular surface disease in AKC is characterized by marked tear film instability, epithelial damage, inflammatory cell infiltration, and increased lipid oxidation(20). Common clinical manifestations of AKC include itching of the eyelids, periorbital region, and conjunctiva; tearing; mucous discharge; burning sensation; photophobia; and blurred vision. Hu et al. demonstrated reduced tear break-up time, corneal sensitivity, and conjunctival goblet cell density, along with increased squamous metaplasia, in patients with AKC(21).
DED is a well-recognized manifestation of AKC, which represents one of the most serious ocular complications of AD. Doğru et al. reported that eyelid eczema, AKC, and superficial punctate keratopathy were the predominant ocular findings in patients with AD. In their study, 62.4% of patients had tear break-up time values below 10 seconds, and 56.2% had Schirmer test values below 5 mm(11). Goblet cell loss and conjunctival squamous metaplasia observed in AKC have also been shown to compromise tear film stability(6). However, whether DED can develop independently of AKC in patients with AD remains unclear, and further studies are required to validate this relationship.
MGs are specialized sebaceous glands that secrete the lipid component of the tear film, thereby reducing tear evaporation. MGD is a chronic, diffuse abnormality of the MGs, typically characterized by terminal duct obstruction and qualitative or quantitative changes in glandular secretion. Because MGD leads to lipid layer abnormalities and tear film instability, MGs play a crucial role in maintaining ocular surface health(22). The pathophysiology of MGD is complex and multifactorial, involving obstructive keratinization of MG orifices, eyelid inflammation, abnormal meibum secretion, and alterations in the ocular surface microbiota(23). MGD is recognized as a major cause of evaporative DED(24). Various inflammatory conditions of the conjunctiva and eyelids, as well as contact lens use, topical or systemic medications, and systemic skin diseases such as rosacea, may disrupt MG structure and function, thereby adversely affecting the ocular surface(25-27).
Several clinical methods have been proposed for the evaluation of MGD, including slit-lamp examination, assessment of tear film lipid layer thickness, tear osmolarity, interferometry, evaporimetry, and meibography. Among these, meibography remains the most widely used technique for clinical evaluation of MG morphology, as recommended by the 2017 MGD Diagnostic Workshop Committee(28). Meibography allows direct in vivo visualization of MG structure, and its diagnostic value for grading MG loss in MGD has been well established(29). However, MG morphology and MGD in newly diagnosed patients with AD have not previously been investigated.
The significant increase in conjunctival redness and MG loss observed in patients with AD in the present study likely reflects inflammation of the periocular skin and mucosa, as well as inflammation-induced vascular changes. Conjunctival redness is an indicator of heightened ocular inflammation and compromised ocular surface barrier function. Chronic inflammation, particularly in patients with AD, is known to induce vascular alterations on the ocular surface, which may exacerbate ocular symptoms. Therefore, increased conjunctival redness may reflect both the severity and persistence of ocular surface inflammation in these patients.
TMH, NITBUT, and the Schirmer test are commonly used parameters for assessing tear film quantity and stability. The significant reductions observed in these parameters, together with the negative correlation between EASI scores and Schirmer test values, suggest that tear film impairment may be present even in the early stages of AD. These alterations are likely attributable to MG dysfunction and inflammatory processes. Furthermore, the higher mean OSDI scores observed in the AD group compared with controls may be explained by reduced tear production and impaired tear quality, leading to symptoms such as ocular dryness, foreign body sensation, and irritation.
To our knowledge, this study is the first to comprehensively evaluate MG morphology and tear film parameters in patients with newly diagnosed AD. Our findings support the notion that patients with AD are predisposed to DED and highlight the association between MG loss and conjunctival inflammation. These results contribute to a better understanding of the pathophysiological mechanisms underlying ocular surface involvement in AD.
Several limitations should be acknowledged. We were unable to assess meibum quality because meibography evaluates only MG morphology, the relatively small sample size, the single-center study design, and the short follow-up duration.
In conclusion, our study demonstrates that ocular surface impairment is present in patients with newly diagnosed AD and that tear film instability may occur at an early stage, even in the absence of AKC. MG loss, increased conjunctival redness, and reduced TMH, NITBUT, and Schirmer test values underscore the potential ocular surface risks associated with AD. These findings suggest that careful monitoring of the tear film and ocular surface is warranted in this patient population, and that early intervention for ocular surface abnormalities may improve patients’ quality of life.
AUTHORS’ CONTRIBUTIONS
Significant contribution to conception and design: Ali Osman Gündogan, Refik Oltulu, Selman Belviranli, Ali Tezcan, Selami Aykut Temiz, Esra Altay. Data acquisition: Ali Osman Gündogan, Selman Belviranli, Ali Tezcan. Data analysis and interpretation: Refik Oltulu, Selami Aykut Temiz, Esra Altay. Manuscript drafting: Ali Osman Gündogan, Refik Oltulu, Selman Belviranli. Significant intellectual content revision of the manuscript: Ali Tezcan, Selami Aykut Temiz, Esra Altay. Final approval of the submitted manuscript: Ali Osman Gündogan, Refik Oltulu, Selman Belviranli, Ali Tezcan, Selami Aykut Temiz, Esra Altay. Statistical analysis: Selman Belviranli. Obtaining funding: not applicable. Supervision of administrative, technical, or material support: Ali Osman Gündogan, Refik Oltulu. Research group leadership: Ali Osman Gündogan, Refik Oltulu.
REFERENCES
1. Weidinger S, Novak N. Atopic dermatitis. Lancet. 2016; 387(10023): 1109-22.
2. Govind K, Whang K, Khanna R, Scott AW, Kwatra SG. Atopic dermatitis is associated with increased prevalence of multiple ocular comorbidities. J Allergy Clin Immunol Pract. 2019;7(1):298-9.
3. Zug KA, Palay DA, Rock B. Dermatologic diagnosis and treatment of itchy red eyelids. Surv Ophthalmol. 1996;40(4):293-306.
4. Dogru M, Okada N, Asano-Kato N, Igarashi A, Fukagawa K, Shimazaki J, et al. Alterations of the ocular surface epithelial mucins 1, 2, 4 and the tear functions in patients with atopic keratoconjunctivitis. Clin Exp Allergy. 2006;36(12):1556-65.
5. Bercovitch L. Screening for ocular complications in atopic dermatitis. Arch Dermatol. 2011;147(5):588-9.
6. Chen JJ, Applebaum DS, Sun GS, Pflugfelder SC. Atopic keratoconjunctivitis: A review. J Am Acad Dermatol. 2014;70(3):569-75.
7. Thyssen JP, Toft PB, Halling-Overgaard AS, Gislason GH, Skov L, Egeberg A. Incidence, prevalence, and risk of selected ocular disease in adults with atopic dermatitis. J Am Acad Dermatol. 2017;77(2):280-286.e1.
8. Knop N, Knop E. [Meibom-Drüsen. Meibomian glands. Part I: anatomy, embryology, and histology of the Meibomian glands]. Ophthalmologe. 2009;106(10):872-83. German.
9. Tomlinson A, Bron AJ, Korb DR, Amano S, Paugh JR, Pearce EI, et al. The international workshop on meibomian gland dysfunction: report of the diagnosis subcommittee. Invest Ophthalmol Vis Sci. 2011;52(4):2006-49.
10. Shimazaki J, Sakata M, Tsubota K. Ocular surface changes and discomfort in patients with meibomian gland dysfunction. Arch Ophthalmol. 1995;113(10):1266-70.
11. Dogru M, Nakagawa N, Tetsumoto K, Katakami C, Yamamoto M. Ocular surface disease in atopic dermatitis. Jpn J Ophthalmol. 1999;43(1):53-7.
12. Mousavi M, Jesus DA, Garaszczuk IK, Szczesna-Iskander DH, Iskander DR. The utility of measuring tear film break-up time for prescribing contact lenses. Cont Lens Anterior Eye. 2018;41(1):105-9.
13. Wu S, Hong J, Tian L, Cui X, Sun X, Xu J. Assessment of bulbar redness with a newly developed keratograph. Optom Vis Sci. 2015;92(8):892-9.
14. Srinivasan S, Menzies K, Sorbara L, Jones L. Infrared imaging of meibomian gland structure using a novel keratograph. Optom Vis Sci. 2012;89(5):788-94.
15. Nutten S. Atopic dermatitis: global epidemiology and risk factors. Ann Nutr Metab. 2015;66(Suppl 1):8-16.
16. Guglielmetti S, Dart JK, Calder V. Atopic keratoconjunctivitis and atopic dermatitis. Curr Opin Allergy Clin Immunol. 2010; 10(5):478-85.
17. Ravn NH, Ahmadzay ZF, Christensen TA, Larsen HH, Loft N, Rævdal P, et al. Bidirectional association between atopic dermatitis, conjunctivitis, and other ocular surface diseases: A systematic review and meta-analysis. J Am Acad Dermatol. 2021;85(2):453-61.
18. Taha RA, Minshall EM, Leung DY, Boguniewicz M, Luster A, Muro S, et al. Evidence for increased expression of eotaxin and monocyte chemotactic protein-4 in atopic dermatitis. J Allergy Clin Immunol. 2000;105(5):1002-7.
19. Wakita H, Sakamoto T, Tokura Y, Takigawa M. E-selectin and vascular cell adhesion molecule-1 as critical adhesion molecules for infiltration of T lymphocytes and eosinophils in atopic dermatitis. J Cutan Pathol. 1994;21(1):33-9.
20. Wakamatsu TH, Dogru M, Ayako I, Takano Y, Matsumoto Y, Ibrahim OM, et al. Evaluation of lipid oxidative stress status and inflammation in atopic ocular surface disease. Mol Vis. 2010;16:2465-75.
21. Hu Y, Matsumoto Y, Dogru M, Okada N, Igarashi A, Fukagawa K, et al. The differences of tear function and ocular surface findings in patients with atopic keratoconjunctivitis and vernal keratoconjunctivitis. Allergy. 2007;62(8):917-25.
22. Nelson JD, Shimazaki J, Benitez-del-Castillo JM, Craig JP, McCulley JP, Den S, et al. The international workshop on meibomian gland dysfunction: report of the definition and classification subcommittee. Invest Ophthalmol Vis Sci. 2011;52(4):1930-7.
23. Bron AJ, de Paiva CS, Chauhan SK, Bonini S, Gabison EE, Jain S, et al. TFOS DEWS II pathophysiology report. Ocul Surf. 2017; 15(3):438-510. Erratum in: Ocul Surf. 2019;17(4):842.
24. Stapleton F, Alves M, Bunya VY, Jalbert I, Lekhanont K, Malet F, et al. TFOS DEWS II Epidemiology Report. Ocul Surf. 2017;15(3):334-65.
25. Arita R, Itoh K, Inoue K, Kuchiba A, Yamaguchi T, Amano S. Contact lens wear is associated with a decrease in meibomian glands. Ophthalmology. 2009;116(3):379-84.
26. Arita R, Itoh K, Maeda S, Maeda K, Furuta A, Tomidokoro A, et al. Effects of long-term topical anti-glaucoma medications on meibomian glands. Graefes Arch Clin Exp Ophthalmol. 2012; 250(8): 1181-5.
27. Palamar M, Degirmenci C, Ertam I, Yagci A. Evaluation of dry eye and meibomian gland dysfunction with meibography in patients with rosacea. Cornea. 2015;34(5):497-9.
28. Wolffsohn JS, Arita R, Chalmers R, Djalilian A, Dogru M, Dumbleton K, et al. TFOS DEWS II Diagnostic Methodology report. Ocul Surf. 2017;15(3):539-74.
29. Arita R, Itoh K, Maeda S, Maeda K, Furuta A, Fukuoka S, et al. Proposed diagnostic criteria for obstructive meibomian gland dysfunction. Ophthalmology. 2009;116(11):2058-63.e1.
Submitted for publication:
September 8, 2025.
Accepted for publication:
December 8, 2025.
Approved by the following research ethics committee: Necmettin Erbakan University (#2024/5152).
Data Availability Statement: The datasets produced and/or analyzed in this study can be provided to referees upon request.
Edited by
Editor-in-Chief: Newton Kara-Júnior
Associate Editor: Dácio C. Costa
Funding: This study received no specific financial support.
Disclosure of potential conflicts of interest: The authors declare no potential conflicts of interest.
How to cite this article: