Arq. Bras. Oftalmol. 2026; 89 (2): 10.5935/0004-2749.2025-0244
Total: 601
Kübra Gündogan1; Ali Osman Gündogan2; Emine Türen Demir1; Naciye Tora2
DOI: 10.5935/0004-2749.2025-0244
ABSTRACT
PURPOSE: Polycystic ovary syndrome is frequently associated with autonomic nervous system dysfunction, even in the absence of obesity or overt metabolic abnormalities. Alterations in pupillary responses may reflect early autonomic involvement and serve as a potential tool for early diagnosis, risk stratification, and disease monitoring. This study aimed to investigate pupillary reflex parameters using dynamic pupillometry in newly diagnosed non-obese women with polycystic ovary syndrome and to compare the findings with those of healthy controls. Methods: This prospective cross-sectional study included 48 newly diagnosed women with polycystic ovary syndrome and 44 age- and sex-matched healthy controls. Pupillary function parameters were measured using dynamic pupillometry (MonPackOne; Metrovision, France). Results: The mean age did not differ significantly between the groups (p=0.870). Initial pupil diameter, pupil contraction amplitude, and contraction velocity were significantly lower in the PCOS group than in the control group, whereas pupillary dilation duration was significantly longer (p<0.001, p<0.001, p=0.007, and p=0.032, respectively). No significant differences were observed between the groups regarding contraction latency, contraction duration, dilation latency, or dilation velocity (p=0.749, p=0.925, p=0.653, and p=0.310, respectively). Conclusion: Newly diagnosed non-obese women with polycystic ovary syndrome exhibit significant alterations in pupillary dynamics, suggesting a generalized reduction in both sympathetic and parasympathetic activity. Dynamic pupillometry may represent a practical, noninvasive tool for detecting early autonomic hypoactivity and identifying patients at risk for future metabolic or cardiovascular complications.
Keywords: Autonomic nervous system; IPupil/physiology; Reflex, Pupillary/physiology; Polycystic ovary syndrome/diagnosis; Menstruation disturbances; Ideal body weight
INTRODUCTION
Polycystic ovary syndrome (PCOS) is a common endocrine disorder affecting women of reproductive age, with a reported prevalence of approximately 6%–10%(1,2). Despite extensive research, its complex pathophysiology has not yet been fully elucidated. Clinical manifestations of PCOS include amenorrhea, oligomenorrhea, hirsutism, obesity, infertility, anovulation, and acne(3). According to the Rotterdam criteria, PCOS is diagnosed when at least two of the following three features are present: ovulatory dysfunction manifested as amenorrhea or oligomenorrhea, clinical and/or biochemical hyperandrogenism, and polycystic ovarian morphology on ultrasonography, after exclusion of other endocrine disorders(4).
The autonomic nervous system (ANS) comprises the sympathetic, parasympathetic, and enteric nervous systems. The enteric nervous system, which primarily regulates gastrointestinal function, acts in coordination with the sympathetic and parasympathetic systems(5). The ANS plays a critical role in the development and progression of PCOS by influencing follicular development, steroid hormone secretion, and glucose and lipid metabolism. Proposed mechanisms include central nervous system activation via afferent pathways, regulation of ovarian blood flow and hormone secretion, direct effects on ovarian function, and modulation of inflammatory and immune responses(6). Several studies have demonstrated the importance of the ANS in ovarian physiology, with increased sympathetic activity reported in women with PCOS(7,8).
Pupillary diameter and light reflex responses are regulated by the ANS through the iris sphincter and dilator muscles. These functions can be objectively and quantitatively assessed using automated pupillometry. In recent years, dynamic pupillometry has been increasingly utilized in the evaluation of autonomic dysfunction(9,10). However, to the best of our knowledge, no studies have examined pupillary light reflex responses using dynamic pupillometry in women with PCOS. Therefore, this study aimed to evaluate pupillary reflex parameters in newly diagnosed non-obese women with PCOS and compare them with those of healthy controls.
METHODS
This cross-sectional clinical study was conducted at the Gynecology and Obstetrics Clinic of Necmettin Erbakan University Faculty of Medicine Hospital between March and June 2025. The study included 48 newly diagnosed, treatment-naive women with PCOS aged 18–45 yr who met the inclusion criteria. Only the right eyes of participants were evaluated. All patients had a body mass index (BMI) <30 kg/m² and were diagnosed according to the 2003 Rotterdam criteria, which include menstrual irregularities (oligomenorrhea or amenorrhea), clinical or biochemical hyperandrogenism, and polycystic ovarian morphology on ultrasonography(11).
Exclusion criteria included endocrine disorders (e.g., diabetes mellitus, Cushing syndrome, and congenital adrenal hyperplasia); systemic diseases (e.g., hypertension, cardiovascular or cerebrovascular disease); gynecological conditions other than PCOS; neurological disorders affecting pupillary function; optic nerve disease; pupillary abnormalities (e.g., synechiae and anisocoria); glaucoma; retinal or corneal disease; history of intraocular surgery or ocular trauma; alcohol or tobacco use; use of systemic medications affecting pupillary function; obesity (BMI ≥30 kg/m²); and a history of COVID-19 infection within the preceding 6 months, confirmed by medical records and participant declaration.
The control group consisted of 44 age- and sex-matched healthy women who attended the same hospital for routine gynecological examination. Inclusion criteria for controls included regular menstrual cycles (21–35 days over the preceding 6 months), absence of clinical or biochemical hyperandrogenism, and normal ovarian morphology on ultrasonography. None of the participants in either group was pregnant, as confirmed by urine β-hCG testing before enrollment.
All participants underwent comprehensive gynecological and ophthalmological examinations, including best-corrected visual acuity assessment, slit-lamp anterior segment examination, fundus examination, and intraocular pressure measurement. All assessments were performed during the follicular phase of the menstrual cycle.
Pupillary light reflex responses were evaluated using dynamic pupillometry (MonPackOne; Metrovision, France). The device uses infrared illumination (880 nm) and a high-resolution sensor to measure pupil parameters in complete darkness. Pupillary responses were elicited using white light stimuli (light intensity: 100 cd/m²; on/off time: 200/3,300 ms) with a measurement precision of 0.1 mm. Measurements were performed between 09:00 and 11:00 to minimize circadian effects. Participants underwent a 5-min dark adaptation period before testing. The following parameters were recorded: initial pupil diameter, pupil contraction amplitude, contraction duration, contraction latency, contraction velocity, dilation latency, dilation velocity, and dilation duration (Figure 1).
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics version 22.0 (IBM Corp., Armonk, New York). Data distribution was assessed using the Kolmogorov–Smirnov and Shapiro–Wilk tests. Group comparisons for normally distributed variables were conducted using the independent samples t test. A p-value <0.05 was considered statistically significant.
RESULTS
The mean age of the 48 patients with PCOS included in the study was 28.08 ± 6.59 yr, while the mean age of the 44 participants in the control group was 27.90 ± 2.52 yr. There was no statistically significant difference between the two groups in terms of mean age (p=0.870; Table 1).
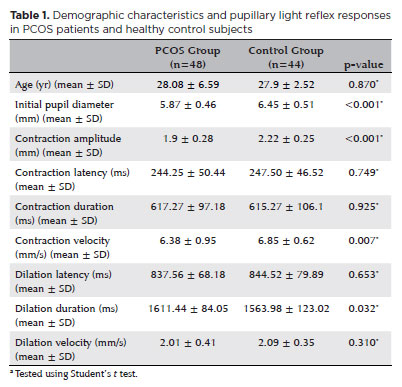
The initial pupil diameter was 5.87 ± 0.46 mm in the PCOS group and 6.45 ± 0.51 mm in the control group. Pupillary contraction amplitude was 1.90 ± 0.28 mm in the PCOS group and 2.22 ± 0.25 mm in the control group, while pupillary contraction velocity was 6.38 ± 0.95 mm/s and 6.85 ± 0.62 mm/s in the PCOS and control groups, respectively. Initial pupil diameter, pupillary contraction amplitude, and contraction velocity were significantly lower in the PCOS group than in the control group (p<0.001, p<0.001, and p=0.007, respectively). Pupillary dilation duration was significantly longer in the PCOS group (1611.44 ± 84.05 ms) than in the control group (1563.98 ± 123.02 ms; p=0.032).
Pupillary contraction latency was 244.25 ± 50.44 ms in the PCOS group and 247.50 ± 46.52 ms in the control group. Pupillary contraction duration was 617.27 ± 97.18 ms and 615.27 ± 106.10 ms, while pupillary dilation latency was 837.56 ± 68.18 ms and 844.52 ± 79.89 ms in the PCOS and control groups, respectively. Pupillary dilation rate was 2.01 ± 0.41 mm/s in the PCOS group and 2.09 ± 0.35 mm/s in the control group. No statistically significant differences were observed between the two groups in terms of pupillary contraction latency, contraction duration, dilation latency, or dilation rate (p=0.749, p=0.925, p=0.653, and p=0.310, respectively; Table 1).
DISCUSSION
In this study, we investigated autonomic dysfunction in newly diagnosed, treatment-naive, non-obese young women with PCOS using dynamic pupillometry. Because obesity independently affects the ANS(12), only non-obese patients were included to more clearly evaluate the direct impact of PCOS on autonomic function. Our findings demonstrated significantly reduced initial pupil diameter, pupillary contraction amplitude, and contraction velocity in the PCOS group compared with healthy controls, along with a significantly prolonged pupillary dilation duration.
The ANS consists of sympathetic and parasympathetic components that play a critical role in maintaining physiological homeostasis. Histological studies have demonstrated ovarian innervation by both sympathetic and parasympathetic (vagal) nerves, including afferent and efferent pathways(13,14). Consequently, the ANS regulates ovarian function by influencing follicular development, steroid hormone secretion, and glucose and lipid metabolism, thereby contributing to the pathophysiology of PCOS(15). Several studies have reported ANS dysfunction in women with PCOS(6,16).
However, findings regarding the nature of autonomic imbalance in PCOS remain inconsistent. Sverrisdóttir et al.(17) reported significantly increased sympathetic nervous system activity in patients with PCOS compared with controls. Similarly, Yu et al.(6), using heart rate variability (HRV) analysis, demonstrated increased sympathetic activity and reduced parasympathetic activity in PCOS patients. In contrast, Di Domenico et al.(8) reported impaired autonomic modulation in response to sympathetic stimulation in young women with the classic PCOS phenotype, a pattern typically observed in older individuals.
Autonomic dysfunction in PCOS has been associated with metabolic and cardiovascular complications, including insulin resistance, hypertension, and increased cardiovascular risk(18,19). Additionally, ANS imbalance may contribute to reproductive dysfunction and hormonal disturbances characteristic of PCOS(20). In the present study, evidence of early autonomic dysfunction was identified in non-obese young women with PCOS, suggesting that cardiovascular and metabolic alterations may begin at an early stage of the disease.
Dynamic pupillometry provides an objective assessment of ANS function by measuring pupillary responses to light stimuli. To the best of our knowledge, this is the first study to evaluate pupillary light reflex parameters in PCOS patients using dynamic pupillometry. Although the systemic effects of PCOS on the ANS are well recognized, its impact on pupillary dynamics has not been adequately investigated.
Quantitative evaluation of the pupillary light reflex enables rapid assessment of both sympathetic and parasympathetic activity. The initial pupil diameter, representing the resting diameter in complete darkness, is primarily regulated by sympathetic tone, whereas pupillary contraction amplitude reflects parasympathetic activity(21). In addition, pupillary dilation parameters predominantly indicate sympathetic nervous system function(22). Wang et al.(23) reported that parasympathetic dysfunction is characterized by prolonged contraction latency and reduced contraction velocity and amplitude.
Previous studies have reported decreased parasympathetic activity in patients with PCOS. Yildirir et al.(24) demonstrated impaired HRV parameters and reduced parasympathetic activity in PCOS patients, while Saranya et al.(25) identified parasympathetic dysfunction through cardiovascular autonomic function tests. Consistent with these findings, the reduced contraction amplitude and velocity observed in our PCOS cohort suggest diminished parasympathetic activity. Moreover, the smaller initial pupil diameter and prolonged dilation duration observed in our study indicate reduced sympathetic activity. Although these findings differ from those reported by Hashim et al.(16) and Yu et al.(6), they are consistent with the observations of Di Domenico et al.(8).
Possible explanations for these discrepancies include differences in study populations, age distribution, body mass index, and metabolic status. Importantly, our study exclusively included newly diagnosed, untreated, and non-obese individuals, suggesting that autonomic dysfunction in early-stage PCOS may be characterized by a generalized reduction in autonomic activity. Increased sympathetic tone observed in later stages of PCOS may be secondary to chronic metabolic disturbances or prolonged hormonal imbalance.
The underlying mechanisms may involve complex neuroendocrine interactions. Dysregulation of the hypothalamic–pituitary–ovarian axis, chronic low-grade inflammation, and insulin resistance may collectively impair central autonomic regulation, leading to reduced autonomic responsiveness. Furthermore, the phenotypic heterogeneity of PCOS may explain divergent autonomic profiles, with sympathetic hyperactivity predominating in obese or metabolically compromised patients, whereas non-obese women in early disease stages may exhibit concurrent reductions in both sympathetic and parasympathetic activity.
This study has several limitations. The sample size was relatively small, and larger studies are required to confirm these findings. Autonomic evaluation was limited to pupillometry, and other ANS assessment methods were not included. Additionally, the single-center, cross-sectional design precluded long-term follow-up, limiting the generalizability of the results. Prospective longitudinal studies are needed to clarify the clinical significance of pupillometric findings in PCOS.
In conclusion, our study demonstrated early autonomic dysfunction in newly diagnosed, treatment-naive, non-obese young women with PCOS using dynamic pupillometry. This dysfunction was characterized by reduced sympathetic and parasympathetic activity. Given the association between autonomic imbalance and metabolic and cardiovascular risk in PCOS, dynamic pupillometry—being noninvasive, rapid, and easily applicable—may serve as a valuable screening tool for early detection of autonomic dysregulation. Early identification of high-risk patients could facilitate timely interventions aimed at preventing long-term complications. Future studies should further explore the pathophysiology and clinical implications of autonomic dysfunction in PCOS.
AUTHORS’ CONTRIBUTIONS
Significant contribution to conception and design: Kübra Gündogan, Ali Osman Gündogan, Emine Türen Demir, Naciye Tora Data acquisition: Ali Osman Gündogan, Kübra Gündogan. Data analysis and interpretation: Emine Türen Demir, Naciye Tora. Manuscript drafting: Ali Osman Gündogan, Naciye Tora. Significant intellectual content revision of the manuscript: Kübra Gündogan, Emine Türen Demir. Final approval of the submitted manuscript: Kübra Gündogan, Ali Osman Gündogan, Emine Türen Demir, Naciye Tora. Statistical analysis: Ali Osman Gündogan. Obtaining funding: not applicable. Supervision of administrative, technical, or material support: Ali Osman Gündogan, Kübra Gündogan. Research group leadership: Ali Osman Gündogan, Kübra Gündogan.
REFERENCES
1. Ehrmann DA. Polycystic ovary syndrome. N Engl J Med. 2005;352(12):1223-36. Comment in: N Engl J Med. 2005; 352(26):2756-7.
2. Azziz R, Woods KS, Reyna R, Key TJ, Knochenhauer ES, Yildiz BO. The prevalence and features of the polycystic ovary syndrome in an unselected population. J Clin Endocrinol Metab. 2004;89(6):2745-9.
3. Chen CH, Wang PH, Hsieh MT, Tzeng CR, Wu YH, Lee CS, et al. Sexual orientations of women with polycystic ovary syndrome: clinical observation in Taiwan. Taiwan J Obstet Gynecol. 2014; 53(4):542-6.
4. Azziz R, Carmina E, Dewailly D, Diamanti-Kandarakis E, Escobar-Morreale HF, Futterweit W, Janssen OE, Legro RS, Norman RJ, Taylor AE, Witchel SF; Task Force on the Phenotype of the Polycystic Ovary Syndrome of The Androgen Excess and PCOS Society. The androgen excess and PCOS Society criteria for the polycystic ovary syndrome: the complete task force report. Fertil Steril. 2009;91(2):456-88.
5. Gibbons CH. Basics of autonomic nervous system function. Handb Clin Neurol. 2019;160:407-18.
6. Yu Y, Chen T, Zheng Z, Jia F, Liao Y, Ren Y, et al. The role of the autonomic nervous system in polycystic ovary syndrome. Front Endocrinol (Lausanne). 2024;14:1295061.
7. Aguado LI. Role of the central and peripheral nervous system in the ovarian function. Microsc Res Tech. 2002;59(6):462-73.
8. Di Domenico K, Wiltgen D, Nickel FJ, Magalhães JA, Moraes RS, Spritzer PM. Cardiac autonomic modulation in polycystic ovary syndrome: does the phenotype matter? Fertil Steril. 2013;99(1):286-92.
9. Aydogmus Y, Uzun S, Gundogan FC, Ulas UH, Ebiloglu T, Goktas MT. Is overactive bladder a nervous or bladder disorder? Autonomic imaging in patients with overactive bladder via dynamic pupillometry. World J Urol. 2017;35(3):467-72.
10. Jackson KG, Malphrus EL, Blum E, Kalloo NB, Finkel JC. Pupillometric assessment of dysautonomia in pediatric bowel and bladder dysfunction: a pilot study. J Pediatr Urol. 2019;15(3):226.e1-226.e5.
11. Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril. 2004;81(1):19-25.
12. Grassi G, Seravalle G, Cattaneo BM, Bolla GB, Lanfranchi A, Colombo M, et al. Sympathetic activation in obese normotensive subjects. Hypertension. 1995;25(4 Pt 1):560-3.
13. Vieyra-Valdez E, Linares-Culebro R, Rosas-Gavilán G, Ramírez-Hernández D, Domínguez-Casalá R, Morales-Ledesma L. Roles of the cholinergic system and vagal innervation in the regulation of GnRH secretion and ovulation: Experimental evidence. Brain Res Bull. 2020;165:129-38.
14. Gerendai I, Tóth IE, Boldogkoi Z, Medveczky I, Halász B. Neuronal labeling in the rat brain and spinal cord from the ovary using viral transneuronal tracing technique. Neuroendocrinology. 1998;68(4):244-56.
15. Uchida S, Kagitani F. Autonomic nervous regulation of ovarian function by noxious somatic afferent stimulation. J Physiol Sci. 2015;65(1):1-9.
16. Hashim ZH, Hamdan FB, Al-Salihi AR. Autonomic dysfunction in women with polycystic ovary syndrome. Iran J Reprod Med. 2015;13(1):27-34.
17. Sverrisdóttir YB, Mogren T, Kataoka J, Janson PO, Stener-Victorin E. Is polycystic ovary syndrome associated with high sympathetic nerve activity and size at birth? Am J Physiol Endocrinol Metab. 2008;294(3):E576-81.
18. Shorakae S, Teede H, de Courten B, Lambert G, Boyle J, Moran LJ. The emerging role of chronic low-grade ınflammation in the pathophysiology of polycystic ovary syndrome. Semin Reprod Med. 2015;33(4):257-69.
19. Esler M, Rumantir M, Wiesner G, Kaye D, Hastings J, Lambert G. Sympathetic nervous system and insulin resistance: from obesity to diabetes. Am J Hypertens. 2001;14(11 Pt 2):304S-309S.
20. Stener-Victorin E, Jedel E, Janson PO, Sverrisdottir YB. Low-frequency electroacupuncture and physical exercise decrease high muscle sympathetic nerve activity in polycystic ovary syndrome. Am J Physiol Regul Integr Comp Physiol. 2009;297(2):R387-95.
21. Yamaji K, Hirata Y, Usui S. A method for monitoring autonomic nervous activity by pupillary flash response. Systems Computers Japan. 2000;31(4):22–31.
22. Micieli G, Tassorelli C, Martignoni E, Marcheselli S, Rossi F, Nappi G. Further characterization of autonomic involvement in multiple system atrophy: a pupillometric study. Funct Neurol. 1995;10(6):273-80.
23. Wang Y, Zekveld AA, Naylor G, Ohlenforst B, Jansma EP, Lorens A, et al. Parasympathetic nervous system dysfunction, as ıdentified by pupil light reflex, and ıts possible connection to hearing ımpairment. PLoS One. 2016;11(4):e0153566.
24. Yildirir A, Aybar F, Kabakci G, Yarali H, Oto A. Heart rate variability in young women with polycystic ovary syndrome. Ann Noninvasive Electrocardiol. 2006;11(4):306-12.
25. Saranya K, Pal GK, Habeebullah S, Pal P. Assessment of cardiovascular autonomic function in patients with polycystic ovary syndrome. J Obstet Gynaecol Res. 2014;40(1):192-9.
Submitted for publication:
August 18, 2025.
Accepted for publication:
November 13, 2025.
Approved by the following research ethics committee: Necmettin Erbakan University (#2025/5586).
Data Availability Statement: The datasets generated and/or analyzed during the current study are already available from the corresponding author upon reasonable request.
Edited by
Editor-in-Chief: Newton Kara-Júnior
Associate Editor: Silvana A. Schellini
Funding: This study received no specific financial support.
Disclosure of potential conflicts of interest: The authors declare no potential conflicts of interest.
How to cite this article: