Arq. Bras. Oftalmol. 2026; 89 (2): 10.5935/0004-2749.2025-0208
Total: 487
Michelle de Lima Farah1; Samir Arbache2; Rubens Belfort Jr.1,3
DOI: 10.5935/0004-2749.2025-0208
ABSTRACT
PURPOSE: To assess the reliability and penetration depth of an automated micropuncture system using a tattoo machine.
METHODS: Twenty human corneas were obtained and subjected to intrastromal micropuncture using a tattoo machine. Each cornea was divided into two halves: one received pigment, while the other received saline solution as a control. The Cheyenne tattoo machine was operated at 60 Hz, with standardized needle exposure (six passes per application). The machine used cartridges containing five microneedles. The study was registered with Agência Nacional de Vigilância Sanitária ANVISA (numbers 80281110015, 80281110016, and 80281110019). The pigment used was Electric Ink black ink, with a density of 1,271,460 μg/mL. Puncture depth was measured before and after the procedure using both anterior segment optical coherence tomography and histopathological analysis. Puncture depth measurements were analyzed using ImageJ software. Each cornea was measured thrice, and the results were subsequently compared.
RESULTS: No corneal perforations were observed with the use of the tattoo machine, and puncture depth measurements ranged from 107 to 486 µm.
CONCLUSIONS: The use of a tattoo machine represents a viable and accessible approach for keratopigmentation, with potential for both cosmetic and therapeutic applications. Its adaptation for controlled intrastromal drug delivery may enable the targeted treatment of deep infectious keratitis, corneal neovascularization, and stromal inflammatory disorders, representing a promising approach for corneal stromal diseases. Further research is needed to optimize techniques and evaluate long-term safety and efficacy, particularly for the delivery of antimicrobial, anti-inflammatory, and anti-vascular endothelial growth factor agents.
Keywords: Eye banks; Cadaver; Cornea; Corneal stroma; Drug delivery systems; Tissue donors; Tattooing/instrumentation; Punctures
INTRODUCTION
The cornea is a compact, avascular structure that serves as the primary refractive surface of the eye. Its unique composition of densely packed collagen fibrils and a highly organized extracellular matrix naturally limits the penetration of both topical medications (eye drops) and systemic medications(1). As a result, achieving effective drug delivery to the corneal stroma remains one of the major challenges in ophthalmology(2).
The challenge of permeating the cornea has fueled the development of novel drug delivery systems capable of overcoming the corneal barrier while maintaining safety and efficacy(3). Some examples include therapeutic contact lenses, femtosecond laser-assisted delivery, and microneedle-based systems(4,5). Despite their advantages, each of these approaches has its limitations, ranging from the need for specialized equipment to concerns regarding ocular toxicity and drug retention. An optimal, widely accessible method for controlled stromal drug delivery is yet to be established.
Dermopigmentation machines (tattoo machines) have emerged as a potential tool for corneal drug delivery. Widely used in dermatology for micropigmentation, these devices create controlled micropunctures that enable the active delivery of pigments or therapeutic substances into the skin(6). Our preliminary investigations confirm that this procedure could be adapted for ophthalmological applications, offering a minimally invasive method for introducing drugs into the corneal stroma(7).
This study aims to evaluate the reliability and penetration depth of an automated micropuncture system using a tattoo machine—a device not previously used for intrastromal punctures in ophthalmology. By measuring variables such as puncture depth reproducibility and potential tissue effects, we assess its potential as a precise and controlled method for corneal applications.
METHODS
Twenty human corneas were used in this experimental study. The study was approved by the Research Ethics Committee of the Federal University of São Paulo (CAAE number: 70612923.7.0000.5505). Each cornea was divided into two halves: one half received an intrastromal pigment injection, while the other received saline solution, both administered using a tattoo machine. The side receiving the pigment or saline solution was randomly assigned to minimize selection bias. The pigment used was Electric Ink black pigment, with a density of 1,271,460 μg/mL.
All corneas were preserved for more than 14 days postmortem. All procedures were performed by a single operator to ensure technical consistency. The volume and pressure of the injection were standardized according to the protocol described in a previous study(8).
The procedure was performed using a Cheyenne tattoo machine and its cartridge (MT.DERM, Berlin, Germany – Agência Nacional de Vigilância Sanitária ANVISA Register: 80281110015 and 80281110019). All interventions were conducted at a needling frequency of 60 Hz, with standardized needle exposure corresponding to six passes, defined as six full rotations of the depth adjustment ring, which controls the projection of the microneedles beyond the cartridge tip. This standardization allowed the evaluation of depth variability under consistent mechanical settings. A sterile cartridge containing five solid microneedles was used for all procedures.
Puncture depth was assessed before and after the procedure by conducting anterior segment optical coherence tomography (OCT) on a Zeiss instrument. All OCT examinations were performed by a single experienced examiner (the first author) under standardized conditions of ambient lighting, bright light, and dark environment. Histopathological analysis was also performed. The puncture measurements were analyzed using ImageJ software (ImageJ 1.54g, Java 1.8.0_345 64-bit). Lateral diffusion measurements were not performed. Each cornea was measured thrice using anterior segment OCT and thrice using histological analysis, and the results were subsequently compared. The reliability of depth measurements obtained through OCT and histological examination was evaluated using the intraclass correlation coefficient (ICC).
Sample size: The inclusion of 20 eyes exceeded the minimum sample required for adequate statistical power. The sample size was estimated using the PASS 14.1 software (Power Analysis and Sample Size System, NCSS, LLC, Kaysville, Utah, USA). Based on pilot data, an ICC of 0.622 for OCT and 0.645 for histology corresponded to powers of 99.6% and 99.8%, respectively, confirming that the adopted sample size was sufficient for the intended analyses.
RESULTS
Moderate reproducibility was observed among the three replicates of OCT measurements and histological measurements, with ICCs of 0.622 (95% confidence interval: 0.381–0.811; p<0.001) and 0.645 (95% confidence interval: 0.410–0.824; p<0.001), respectively.
OCT analysis demonstrated mean stromal depths of 234.80 µm (standard deviation [SD]=62.19 µm), 238.15 µm (SD=61.07 µm), and 224.65 µm (SD=50.62 µm) across the three replicates. The minimum and maximum depths measured were 107 µm and 410 µm, respectively. No corneal perforation was identified, indicating that all micropunctures remained confined within the stromal layer.
Histological evaluation revealed mean depths of 210.90 µm (SD=77.25 µm), 203.90 µm (SD=102.19 µm), and 188.85 µm (SD=72.07 µm) across the three replicates, with minimum and maximum values of 94 µm and 481 µm, respectively. Histological measurements exhibited greater variability than OCT measurements.
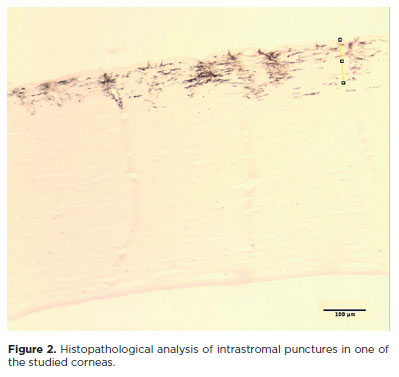
Overall, the stromal depths obtained by OCT were slightly greater than those measured histologically. Both methods yielded mean values of depth within the expected range for intrastromal micropunctures (150–250 µm), demonstrating consistency between optical and histological assessments. The depth of stromal puncture varied from 107 µm to 481 µm (Figures 1 and 2). In addition, pachymetry analysis performed by OCT before and after the procedure revealed an average increase in corneal thickness of 31.7 ± 50.5 µm (p=0.011). No transfixation was observed in any of the corneas analyzed, as confirmed by both OCT and histopathological examination.
DISCUSSION
Pigments or other substances can be delivered into the cornea manually or via an automated system(7). While the micropuncture system provides improved control over needle exposure, drug delivery remains technique-dependent, with inherent variability in all intrastromal needle injection methods due to factors such as corneal properties, applied pressure, injected substance density, manual measurement techniques, learning curve, and cartridge reuse. In this study, OCT and histological measurements demonstrated moderate reproducibility, indicating acceptable agreement but also some unavoidable variability. These fluctuations likely result from a combination of mechanical and biological factors, including minor shifts in measurement positioning and operator-dependent variations during data acquisition. In addition, OCT readings may be affected by image resolution and segmentation accuracy, whereas histological evaluations can be influenced by tissue processing artifacts and sectioning inconsistencies.
The differences observed between OCT and histological depth measurements are consistent with previous reports describing mild discrepancies between in vivo optical imaging and ex vivo histological evaluations. This divergence is mainly attributed to stromal shrinkage during histological processing—particularly during fixation, dehydration, and paraffin embedding—which can reduce apparent tissue thickness. Minor differences in sectioning planes or image alignment may further contribute to the observed variability(9,10). Despite these variations, both methodologies demonstrated mean depths within the optimal range for safe intrastromal injections (150–250 µm), validating the depth control and reproducibility achieved by the automated system.
In superficial anterior keratopigmentation, pigment deposition typically occurs at a stromal depth of 120–150 µm, providing long-term stability while avoiding epithelial irritation and deeper stromal injury(11,12). In our study, most of the punctures remained within the 150–250 µm range, which is considered optimal for effective intrastromal delivery, although isolated deeper penetrations up to 481 µm were also observed(12-14). These findings suggest that although deeper injections are technically feasible, the critical factor for achieving effective outcomes is successful intrastromal delivery without corneal perforation, rather than minor variations in depth.
Previous studies have used automated platforms for keratopigmentation, but this is the first study to employ the Cheyenne tattoo machine for corneal micropuncture applications. Alió et al. developed the Vissum Eye MP system (Blue Green Medical, Madrid, Spain), an ophthalmic-specific device that demonstrated high precision, reproducibility, and minimal tissue trauma during pigment delivery(11-13). Our results indicate that the Cheyenne machine, despite being a commercially available nonophthalmic device, can achieve reliable and controlled intrastromal micropunctures when operated under standardized parameters and with appropriate training. Both anterior segment OCT and histological analyses showed moderate reproducibility in depth measurements, with most punctures confined to the optimal stromal range, demonstrating that the system enables safe and effective intrastromal deposition. These results highlight its potential as a practical and accessible alternative in clinical keratopigmentation and experimental corneal drug delivery applications.
Building on these findings, emerging microneedle-based platforms in corneal drug delivery seek to replicate and further enhance the precision and control achieved by automated intrastromal micropuncture systems. Although dissolving microneedle technologies for corneal application are not yet available in clinical practice, recent experimental studies have shown promising results. Compared with conventional topical or intrastromal injection methods, polymeric microneedles have demonstrated superior drug bioavailability and therapeutic efficacy in preclinical models of anterior segment disorders, including infection, dystrophy, and neovascularization(14,15). Microneedle patches have been used to successfully deliver anti-vascular endothelial growth factor and anti-inflammatory agents directly into the stroma, effectively reducing neovascularization and accelerating corneal healing(16). Although innovations such as bilayer and annular microneedle designs remain in the experimental stage pending regulatory approval for human use, they have demonstrated the ability to enhance the safety, precision, and control of stromal drug delivery(17).
An important limitation of the present study is the absence of lateral diffusion analysis. Understanding the lateral spread of pigments or drugs within the corneal stroma is critical for evaluating both therapeutic efficacy and safety, particularly in clinical applications where precise localization of the substance is required. Our current methodology has focused on assessing intrastromal depth and reproducibility. Future studies have been planned to quantify lateral dispersion and the actual volume of pigment or drug deposited. Such investigations will provide valuable insights into optimizing dosing, minimizing unintended diffusion, and ensuring consistent outcomes in both experimental and clinical settings.
Future work will focus on in vivo testing, comprehensive evaluation of inflammatory and functional responses, and controlled clinical trials to determine the translational applicability of this approach in patients, ultimately supporting the potential of automated intrastromal micropuncture as a reliable and versatile technique for both therapeutic and cosmetic corneal applications.
ACKNOWLEDGMENTS
This study was supported by CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior), process number 001, and institutional support from Instituto da Visão (IPEPO) and the Department of Ophthalmology and Visual Sciences and the Postgraduate Program in Ophthalmology and Visual Sciences of Escola Paulista de Medicina (Federal University of São Paulo — UNIFESP).
AUTHORS’ CONTRIBUTIONS:
Significant contribution to conception and design: Rubens Belfort Jr. Data acquisition: Michelle de Lima Farah. Data analysis and interpretation: Michelle de Lima Farah, Samir Arbache. Manuscript drafting: Michelle de Lima Farah, Samir Arbache, Rubens Belfort Jr. Significant intellectual content revision of the manuscript: Michelle de Lima Farah, Samir Arbache, Rubens Belfort Jr. Final approval of the submitted manuscript: Michelle de Lima Farah, Samir Arbache, Rubens Belfort Jr. Statistical analysis: Michelle de Lima Farah, Samir Arbache. Obtaining funding: Michelle de Lima Farah, Rubens Belfort Jr. Supervision of administrative, technical, or material support: Michelle de Lima Farah, Samir Arbache. Research group leadership: Rubens Belfort Jr.
REFERENCES
1. Michelacci YM. Collagens and proteoglycans of the corneal extracellular matrix. Braz J Med Biol Res. 2003;36(8):1037-46.
2. Massoudi D, Malecaze F, Galiacy SD. Collagens and proteoglycans of the cornea: importance in transparency and visual disorders. Cell Tissue Res. 2016;362(2):337-49.
3. Abdi B, Mofidfar M, Hassanpour F, Kirbas Cilingir E, Kalajahi SK, Milani PH, et al. Therapeutic contact lenses for the treatment of corneal and ocular surface diseases: advances in extended and targeted drug delivery. Int J Pharm. 2023;638:122740.
4. Bhatnagar S, Saju A, Cheerla KD, Gade SK, Garg P, Venuganti VV. Corneal delivery of besifloxacin using rapidly dissolving polymeric microneedles. Drug Deliv Transl Res. 2018;8(3):473-83.
5. Thakur RR, Tekko IA, Al-Shammari F, Ali AA, McCarthy H, Donnelly RF. Rapidly dissolving polymeric microneedles for minimally invasive intraocular drug delivery. Drug Deliv Transl Res. 2016;6(6):800-15.
6. Arbache S, de Mendonça MT, Arbache ST, Hirata SH. Treatment of idiopathic guttate hypomelanosis with a tattoo device versus a handheld needle. JAAD Int. 2021;3:14-6.
7. Farah M de L, Arbache S, Grupenmacher AT, Muralha F, Lima Filho AA, Maia M, et al. Corneal drug delivery with tattoo technique: A new route for intrastromal cornea administration. Pan Am J Ophthalmol. 2023;5(1):51.
8. Arbache S, Mattos E da C, Diniz MF, Paiva PY, Roth D, Arbache ST, et al. How much medication is delivered in a novel drug delivery technique that uses a tattoo machine? Int J Dermatol. 2019;58(6):750-5.
9. Alonso-Caneiro D, Read SA, Vincent SJ, Collins MJ, Wojtkowski M. Tissue thickness calculation in ocular optical coherence tomography. Biomed Opt Express. 2016;7(2):629-45.
10. Patel A, Cholkar K, Agrahari V, Mitra AK. Ocular drug delivery systems: an overview. World J Pharmacol. 2013;2(2):47-64.
11. Hasani H, Es’haghi A, Rafatnia S, Alilou S, Abolmaali M. Keratopigmentation: a comprehensive review. Eye (Lond). 2020;34(6):1039-46.
12. Alio JL, Amesty MA, Rodrigues A, El Bahrawy M. Text and atlas on corneal pigmentation. Nova Deli, Jaypee Brothers Medical; 2015.
13. Rodriguez AE, Amesty MA, El Bahrawy M, Rey S, Alio del Barrio J, Alio JL. Superficial automated keratopigmentation for iris and pupil simulation using micronized mineral pigments and a new puncturing device: experimental study. Cornea. 2017;36(9):1069-75.
14. Shi H, Zhou J, Wang Y, Zhu Y, Lin D, Lei L, et al. A rapid corneal healing microneedle for efficient ocular drug delivery. Small. 2022;18(4):e2104657.
15. Glover K, Mishra D, Gade S, Vora LK, Wu Y, Paredes AJ, et al. Microneedles for advanced ocular drug delivery. Adv Drug Deliv Rev. 2023;201:115082.
16. Mulkutkar M, Damani M, Sawarkar S. Polymeric microneedles for the eye: an overview of advances and ocular applications for minimally invasive drug delivery. Eur J Pharm Biopharm. 2024;197:114209.
17. Li L, Jia F, Wang Y, Liu J, Tian Y, Sun X, et al. Transcorneal drug delivery strategies in the treatment of ocular diseases. Adv Drug Deliv Rev. 2023;198:114868.
Submitted for publication:
July 18, 2025.
Accepted for publication:
December 8, 2025.
Approved by the following research ethics committee: Universidade Federal de São Paulo – UNIFESP (CAAE: 70612923.7.0000.5505).
Data Availability Statement:
The datasets generated and/or analyzed during the current study are included in the manuscript.
Edited by
Editor-in-Chief: Newton Kara-Júnior
Associate Editor: Dácio C. Costa
Disclosure of potential conflicts of interest: The authors declare no potential conflicts of interest.
How to cite this article: