

Caio Costa Santos; Roberta Tovo Borghetto Abud; Alvio Isao Shiguematsu; Daniela Carvalho dos Santos
DOI: 10.5935/0004-2749.2025-0105
ABSTRACT
PURPOSE: To evaluate structural differences in amniotic membrane fragments subjected to different preservation techniques for potential ophthalmologic applications.
METHODS: Three placentas were collected from healthy donors, and four amniotic membrane fragments were prepared from each placenta. The fragments were divided into four groups with three samples each: cryopreserved, lyophilized, vacuum-dried using a vacuum concentrator, and fresh (control). After processing, the fragments were fixed, sectioned, and analyzed using scanning transmission electron microscopy to assess tissue morphology.
RESULTS: All samples met the established evaluation criteria. No morphological differences were observed among the groups. The structural characteristics of lyophilized and vacuum-dried membranes were comparable with those of cryopreserved and fresh membranes. However, vacuum drying demonstrated the greatest practicality for ophthalmologic use, as it allows membrane availability at any time and storage at room temperature.
CONCLUSION: Vacuum drying using a vacuum concentrator, lyophilization, and cryopreservation preserve the morphological characteristics of the human amniotic membrane similar to those of fresh tissue. A standardized protocol using a vacuum concentrator may be established owing to its advantages in storage convenience and accessibility.
Keywords: Amnion/transplantation; Cryopreservation/methods; Freeze drying; Lyophilization; Ophthalmologic surgical procedures; Regenerative medicine
INTRODUCTION
The human amniotic membrane (HAM) has multiple applications in regenerative medicine. It functions as a three-dimensional framework rich in various types of collagen, acting as a highly biocompatible natural scaffold and a source of stem cells and potent growth factors. HAM fragments can also serve as effective nano-reservoirs for drug delivery due to their high capacity to trap different molecules. Clinical applications range from simple sheet or topical use for skin and corneal repair to more advanced forms, such as micronized dehydrated membranes, amniotic cytokine extracts, and injections of solubilized powder for tissue regeneration(1-3).
The amniotic membrane is the innermost of the three fetal membranes. It is an avascular structure composed of five cellular layers and features a basement membrane similar to that of the conjunctiva(2). The first use of HAM in ophthalmology was reported in 1940, for the repair of conjunctival defects(3,4). The amniotic membrane contains numerous growth factors, including interleukin-10 (IL-10), platelet-derived growth factors (PDGF-AA and PDGF-BB), basic fibroblast growth factor (FGFβ), epidermal growth factor, keratinocyte growth factor, keratinocyte growth factor receptor, hepatocyte growth factor, hepatocyte growth factor, transforming growth factor-β (TGFβ), tissue inhibitors of metalloproteinases (TIMP-1, TIMP-2, TIMP-3, and TIMP-4), and vascular endothelial growth factor (VEGF). These factors contribute to HAM’s ability to promote healing, epithelialization, and anti-inflammatory effects(2,4,5). In addition, HAM contains bactericidin, β-lysine, lysozyme, transferrin, and immunoglobulins, which provide antimicrobial protection. Its low immunogenicity further supports its utility as a graft material(6).
The use of HAM in ophthalmology has grown in recent years, with reports of its application in ocular surface reconstruction, pterygium surgery, corneal perforations, persistent epithelial defects, infectious keratitis, and ocular burns(5).
The selection of placenta donors and the preparation of HAM are complex processes that require strict control. Several preservation techniques have been described. The most widely used worldwide is cryopreservation with glycerol, which involves storage at −80°C. However, this method requires costly and specialized freezers that are often not widely available(2,7). Lyophilization, a tissue dehydration process through sublimation, preserves cytokine levels and the physical strength of the membrane compared with fresh tissue, while also allowing storage at room temperature(8,9). Favorable results with lyophilized HAM have been reported in pterygium surgery(10). A newer alternative, vacuum drying using a vacuum concentrator, has been described but remains sparsely discussed in the literature.
This study aimed to evaluate the morphological structure of HAM using scanning electron microscopy (SEM) and transmission electron microscopy (TEM) following different preservation methods—fresh, cryopreserved at −80°C, vacuum-dried using a vacuum concentrator, and lyophilized—to identify the most suitable preservation method and to establish a protocol for vacuum concentrator-based HAM preservation.
METHODS
This experimental study was conducted to determine the most appropriate protocol for HAM preservation for ophthalmologic applications. All procedures were performed in accordance with the guidelines of the local research ethics committee.
Amniotic membranes were obtained from donors aged 18-36 yr who met the following inclusion criteria: at least two prenatal consultations, gestational age ≥35 weeks, normal labor without complications, cesarean delivery, no use of drugs that could be deposited in tissues, no diseases of the genital tract or systemic diseases posing risks to recipients, and no personal or family history of hereditary disorders. Only donors with negative tests for HIV, hepatitis B and C, HTLV I/II, and syphilis were included. The placenta was examined macroscopically and microscopically to exclude neoplasms. Eligible fetuses weighed ≥2,000 g, had no congenital abnormalities, and had no history of severe fetal distress.
After obtaining informed consent, the placenta was placed in a sterile container and transported to the tissue bank under refrigerated conditions (below 8°C). In a sterile environment, the placenta was washed with saline to remove clots and excess blood. A cut was made near the umbilical cord, and the amniotic membrane was separated from the chorion by blunt dissection. Large amniotic membrane pieces were collected and rinsed with saline to eliminate residual blood. Transparent and uniformly thick amniotic membrane segments were selected and washed with an antibiotic solution containing penicillin (10,000 IU/mL), ceftazidime (10 mg/mL), and amphotericin B (25 mg/mL).
The HAM was then mounted on a nitrocellulose carrier with the epithelial side facing upward and cut into 2 × 2 cm sections. Placentas were obtained from three donors, yielding one sample each of fresh, cryopreserved, lyophilized, and vacuum-dried HAM for morphological analysis (total: 12 samples).
For cryopreservation, amniotic membrane segments were placed in vials containing a glycerol solution and Dulbecco’s Modified Eagle’s Medium in a 1:1 ratio and stored in a freezer at −80°C. Under this preservation method, the shelf life of the preserved amniotic membrane is up to 2 yr. Before use, the membrane was allowed to reach room temperature and thawed for 3-5 min, after which it was suitable for ophthalmological application.
For vacuum drying using a vacuum concentrator, immediately after preparing the amniotic membrane fragments, the samples were placed in a fenestrated container and positioned inside a concentrator equipped with an integrated SpeedVac 230 V/50-60-Hz diaphragm vacuum pump (Eppendorf) to dry the membrane. The samples were processed at 45°C for 24 h. Upon removal, the membranes were completely dehydrated and remained stable at room temperature. For use, each sample was rehydrated by applying five drops of distilled water and left to soak for 5 min. The rehydrated amniotic membrane appeared light yellow, semi-translucent, and slightly less elastic than the fresh membrane.
Lyophilization, a tissue dehydration process achieved through sublimation, was performed using a lyophilizer (Alpha 2-4 LDplus, Martin Christ, Germany, 2017). Amniotic membrane fragments were first frozen in an ultrafreezer at −80°C for 30 min and then processed in the lyophilizer for 24 h. The samples were removed once a dry macroscopic appearance was achieved and remained stable at room temperature. For use, the samples were rehydrated with five drops of distilled water and allowed to stand for 5 min. The macroscopic appearance of both vacuum-dried and lyophilized samples was very similar before and after hydration.
After preparation, both the vacuum-dried and lyophilized samples were rehydrated, cut into small fragments, and placed in tubes containing fixative for slide preparation and evaluation. The samples were analyzed for the preservation of cellular morphology and extracellular matrix integrity using SEM and TEM. To ensure objective evaluation, six indicators were defined for TEM-based comparison of amniotic membrane preserved by different methods versus fresh (in natura) samples: (1) intact basement membrane; (2) intact collagen layer; (3) intact cell nucleus with visible double membrane; (4) presence of desmosomes and intermediate filaments in the cytoplasm; (5) preservation of apical projections and villi; and (6) presence of organelles or cytoplasmic vesicular complexes.
SEM was used to assess the structure and spatial relationship between the collagen, epithelial, and basement membrane layers, comparing the different preservation groups with the fresh membrane.
As described, four study groups were established for structural analysis of the amniotic membrane based on the preservation method: Group 1: fresh amniotic membrane (n=3); Group 2: cryopreserved amniotic membrane (n=3); Group 3: lyophilized amniotic membrane (n=3); Group 4: vacuum-dried amniotic membrane using a vacuum concentrator (n=3).
RESULTS
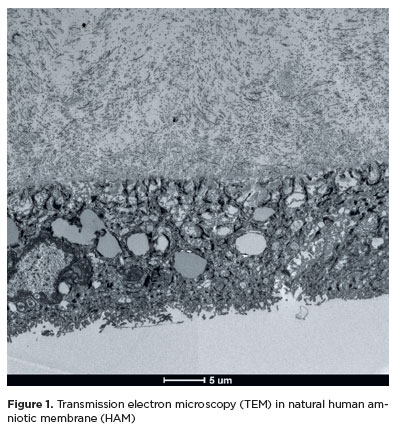
In all fresh samples, epithelial cells with irregular shapes were observed, featuring apical projections or villi and surface invaginations, a thin basement membrane, and abundant collagen arranged in varied orientations. The cytoplasm of the epithelial cells contained numerous intermediate filaments and desmosomes, ensuring cell adhesion, multiple lipid-containing vesicles, and nuclei with double envelopes. Some cells also exhibited multivesicular complexes near the nucleus.
As shown in figure 1, the epithelium and a large quantity of collagen were evident.
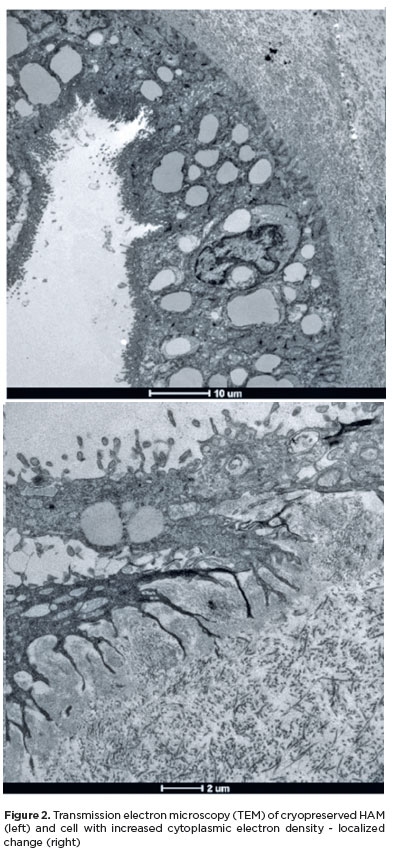
All samples from the cryopreserved HAM group displayed an intact basement membrane, nuclei with double envelopes, an extensive collagen layer (Figure 2), organelles near the nucleus, and numerous intermediate filaments and desmosomes as well as preserved apical villi. The cryopreserved HAM sample from placenta 3 exhibited an epithelial cell with increased cytoplasmic electron density (Figure 2); however, this alteration was localized, while the remaining cells showed good preservation characteristics.
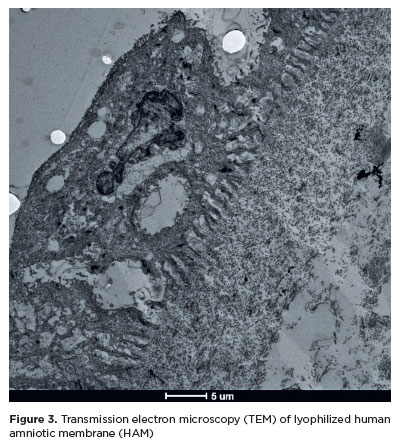
The lyophilized HAM also retained the morphological features of fresh HAM (Figure 3), differing only by a slight increase in the number of lipid vesicles (Figure 4). The basal membrane and collagen layer were preserved, organelles and nuclei remained intact, and multiple desmosomes, intermediate filaments, and apical villi were observed.
HAM samples subjected to vacuum drying using a concentrator also demonstrated good structural preservation (Figure 5). The nuclei exhibited chromatin marginalization and irregular shapes but no signs of programmed cell death. The double nuclear envelope, thick collagen layer, intact basement membrane, cytoplasmic organelles, preserved apical villi, and abundant intermediate filaments and desmosomes were maintained.
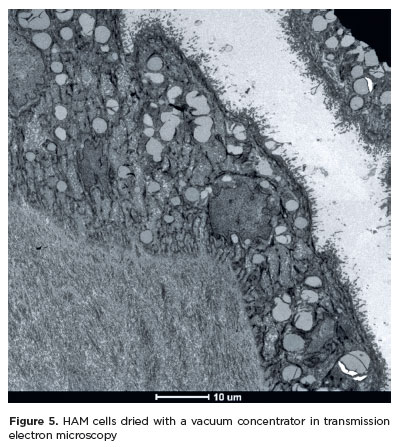
Table 1 and figure 6 summarize the findings of the TEM analysis.
Structural assessment using SEM showed that fresh HAM consisted of epithelial cells overlying a thick collagen layer with fibers arranged in multiple directions. The apical projections (villi) were visible on the epithelial cell surfaces. Samples preserved by cryopreservation, lyophilization, and vacuum drying maintained the same tissue architecture as fresh HAM, without areas of fragmentation, epithelial denudation, or extracellular matrix decompaction. Figure 7 presents representative SEM images from each preservation group, showing a single epithelial cell layer over a dense, multi-directional collagen network (top) and intact apical villi on the epithelial surface (bottom).
DISCUSSION
The amniotic membrane consists of a simple epithelium of cuboidal or columnar cells with apical microvilli(11). In this study, preservation of the epithelial layer and microvilli was observed following lyophilization, cryopreservation, and vacuum drying. Two other layers—the basement membrane and the stroma, rich in collagen—were also well preserved(11).
The HAM possesses distinctive features that make it highly suitable for ophthalmic applications. It provides structural support, facilitates epithelialization and corneal and conjunctival healing, reduces inflammation, and has low immunogenicity(12). Additionally, it offers better cosmetic outcomes than oral or mucosal grafts, which typically result in non-conjunctival epithelial morphology(11). HAM epithelial cells are known to secrete pigment-derived growth factors, neurotransmitters, neuropeptides, and neurotrophic factors that contribute to their therapeutic potential in ophthalmology(2). The epithelial cells in our study retained intact nuclei and organelles following vacuum drying, cryopreservation, and lyophilization, suggesting that after rehydration or thawing, HAM epithelial cells may remain functional—although this hypothesis was beyond the scope of the present study.
HAM can be used on the ocular surface either as a permanent graft, integrating into the host tissue, or as a biological dressing that alleviates pain and reduces inflammation(12). When used as a graft, it is applied with the epithelial side facing upward on the wound bed(11). The HAM basement membrane closely resembles that of the conjunctiva and cornea, serving as a substrate for epithelial cell growth on the ocular surface(5,11). Moreover, HAM forms desmosomes and hemidesmosomes, which facilitate its integration into the ocular stroma(11). Therefore, preservation techniques that maintain an intact basement membrane and a high density of desmosomes are essential. In this study, all three methods—vacuum drying, lyophilization, and cryopreservation—preserved both features effectively.
When used in ophthalmology, inflammatory cells become trapped within the HAM stroma, where they undergo apoptosis, preventing the initiation of inflammatory cascades on the ocular surface(11,13). The collagen-rich stroma, which plays a key role in this anti-inflammatory action, remained intact in all samples. HAM can be conveniently applied in-office under a therapeutic contact lens for cases such as corneal ulcers, neurotrophic keratopathy, or chemical burns, and it can be used concurrently with topical medications(14). In bacterial keratitis, HAM offers additional benefits: it does not impede antibiotic penetration and can absorb antimicrobial agents, thereby prolonging their therapeutic effect(12).
For clinical use, it is logistically advantageous to employ HAM that can be stored at room temperature, enabling ophthalmologists to keep it readily available for immediate application. Thus, lyophilized and vacuum-dried HAM have practical advantages over cryopreserved HAM. Vacuum-dried HAM, in particular, offers operational benefits, requiring only a single device for processing, whereas lyophilization necessitates a preliminary freezing step at −80°C before lyophilization. Furthermore, the vacuum concentrator is a compact, lightweight, and easily transportable device, allowing for HAM processing in the same environment where childbirth occurs, if desired and planned by the medical team.
In conclusion, under the experimental conditions of this study, vacuum drying, lyophilization, and cryopreservation all maintained the morphological characteristics of HAM comparable with those of fresh tissue. However, vacuum drying using a concentrator represents a more practical and logistically favorable preservation method.
AUTHORS’ CONTRIBUTIONS:
Significant contribution to conception and design: Caio Costa Santos, Roberta Tovo Borghetto Abud. Data acquisition: Caio Costa Santos, Alvio Isao Shiguematsu, Daniela Carvalho dos Santos. Data analysis and interpretation: Caio Costa Santos, Alvio Isao Shiguematsu, Daniela Carvalho dos Santos. Manuscript drafting: Caio Costa Santos, Roberta Tovo Borghetto Abud. Significant revision of the intellectual content of the manuscript: Caio Costa Santos, Roberta Tovo Borghetto Abud. Final approval of the submitted manuscript: Caio Costa Santos, Roberta Tovo Borghetto Abud, Alvio Isao Shiguematsu, Daniela Carvalho dos Santos. Statistical analysis: Caio Costa Santos. Obtaining funding: not applicable. Supervision of administrative, technical, or material support: Caio Costa Santos. Research group leadership: Caio Costa Santos.
REFERENCES
1. Elkhenany H, El-Derby A, Abd Elkodous M, Salah RA, Lotfy A, El-Badri N. Applications of the amniotic membrane in tissue engineering and regeneration: the hundred-year challenge. Stem Cell Res Ther. 2022;13(1):1-19.
2. Ferenczy PA, Souza LB. Comparison of the preparation and preservation techniques of amniotic membrane used in the treatment of ocular surface diseases. Rev Bras Oftalmol. 2020;79(1):71-80.
3. Rotth A. Plastic repair of conjunctival defects with fetal membranes. Arch Ophthalmol. 1940;23(3):522-5.
4. Dua HS, Gomes JA, King AJ, Maharajan VS. The amniotic membrane in ophthalmology. Surv Ophthalmol. 2004;49(1):51-77.
5. Jirsova K, Jones GL. Amniotic membrane in ophthalmology: properties, preparation, storage and indications for grafting—a review. Cell Tissue Bank. 2017;18(2):193-204.
6. Leal‐Marin S, Kern T, Hofmann N, Pogozhykh O, Framme C, Börgel M, et al. Human amniotic membrane: a review on tissue engineering, application, and storage. J Biomed Mater Res B Appl Biomater. 2021;109(8):1198-215.
7. Paolin A, Trojan D, Leonardi A, Mellone S, Volpe A, Orlandi A, et al. Cytokine expression and ultrastructural alterations in fresh-frozen, freeze-dried and γ-irradiated human amniotic membranes. Cell Tissue Bank. 2016;17(3):399-406.
8. Nakamura T, Yoshitani M, Rigby H, Fullwood NJ, Ito W, Inatomi T, et al. Sterilized, freeze-dried amniotic membrane: a useful substrate for ocular surface reconstruction. Invest Ophthalmol Vis Sci. 2004;45(1):93-9.
9. Nakamura T, Inatomi T, Sekiyama E, Ang LP, Yokoi N, Kinoshita S. Novel clinical application of sterilized, freeze-dried amniotic membrane to treat patients with pterygium. Acta Ophthalmol Scand. 2006;84(3):401-5.
10. Allen CL, Clare G, Stewart EA, Branch MJ, McIntosh OD, Dadhwal M, et al. Augmented dried versus cryopreserved amniotic membrane as an ocular surface dressing. PLoS One. 2013;8(10):e007.
11. Sharma R, Nappi V, Empeslidis T. The developments in amniotic membrane transplantation in glaucoma and vitreoretinal procedures. Int Ophthalmol. 2023;43(5):1771-83.
12. Röck T, Bartz-Schmidt KU, Landenberger J, Bramkamp M, Röck D. Amniotic membrane transplantation in reconstructive and regenerative ophthalmology. Ann Transplant. 2018;23:160-5.
13. Shimmura S, Shimazaki J, Ohashi Y, Tsubota K. Anti-inflammatory effects of amniotic membrane transplantation in ocular surface disorders. Cornea. 2001;20(4):408-13.
14. McGaughy AG, Gupta PK. In-office use of amniotic membrane. Ophthalmic Pearls Cornea. 2015.
Submitted for publication:
May 12, 2025.
Accepted for publication:
October 8, 2025.
Approved by the following research ethics committee: Faculdade de Medicina de Botucatu – UNESP (CAAE: 75897023.0.0000.5411).
Data Availability Statement: The datasets generated and/or analyzed during the current study are included in this manuscript.
Edited by
Editor-in-Chief: Newton Kara-Júnior
Associate Editor: Richard Y. Hida
Funding: This study received no specific financial support.
Disclosure of Potential Conflicts of Interest: The authors declare no potential conflicts of interest.