Arq. Bras. Oftalmol. 2024; 87 (4): 10.5935/0004-2749.2023-0026
Total: 2640
Allexya Affonso Antunes Marcos1,2; Denise Freitas1; Jeison de Nadai Barros1; Arthur Gustavo Fernandes1; Márcia Lowen2; Moacyr Rigueiro2; Melina Correia Morales1; Rubens Belfort Neto1; Arun D. Singh3
DOI: 10.5935/0004-2749.2023-0026
ABSTRACT
PURPOSE: To describe cellular alterations detected by impression cytology of the ocular surface in patients with xeroderma pigmentosum. The secondary objective was to assess the reliability of impression cytology in diagnosing ocular surface squamous neoplasia.
METHODS: Patients with xeroderma pigmentosum underwent a single-day complete ophthalmological examination and impression cytology for ocular surface evaluation using 13 mm diameter mixed cellulose esters membrane filters and combined staining with Periodic Acid Schiff, Hematoxylin and Eosin, and Papanicolaou stains followed by microscopic analysis. The cytological findings were correlated with the clinical diagnosis. The impression cytology findings at baseline and one-year follow-up were correlated with the clinical course (no tumor, treated tumor, residual tumor recurrent tumor, new tumor).
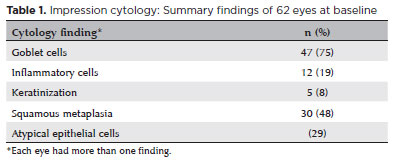
RESULTS: Of the 42 patients examined, impression cytology was performed in 62 eyes of 34 participants (65% females). The mean age of patients was 29.6 ± 17 years (range 7-62). Fifteen eyes had a clinical diagnosis of ocular surface squamous neoplasia. Impression cytology showed goblet cells (47, 75%), inflammatory cells (12, 19%), keratinization (5, 8%), and squamous metaplasia (30, 48%). Impression cytology was positive for atypical cells in 18 patients (12 with and 6 without ocular surface squamous neoplasia). The sensitivity, specificity, positive predictive value, and negative predictive value of impression cytology (at baseline) for diagnosis of ocular surface squamous neoplasia were 80%, 87%, 67%, and 93%, respectively, using clinical diagnosis of ocular surface squamous neoplasia as the reference standard.
CONCLUSION: Impression cytology has a moderate positive predictive value for the diagnosis of ocular surface squamous neoplasia in patients with xeroderma pigmentosum. However, the lack of detection of atypical cells on impression cytology has a high negative predictive value for ocular surface squamous neoplasia. Integration of impression cytology in the long-term management of high-risk patients, such as patients with xeroderma pigmentosum, can avoid unnecessary diagnostic biopsies.
Keywords: Xeroderma pigmentosum; Eye neoplasms; Conjunctiva/cytology; Cornea/cytology; Cytological techniques
INTRODUCTION
Xeroderma pigmentosum (XP) is a rare type of genodermatosis inherited as an autosomal recessive condition. It is characterized by hypersensitivity to sunlight resulting in exaggerated photoaging and photocarcinogenesis(1-3). Compared to the general population, XP patients aged up to 20 years have a 1000-fold higher risk of cancer of the sun-exposed tissues of the eye(2).
Impression cytology (IC) is a technique for sampling the superficial layers of the ocular surface by applying collecting devices. Cells adhering to the surface of the device can be removed and processed further for analysis by a variety of methods. It is a simple and noninvasive technique for diagnosis and posttreatment follow-up of several ocular surface disorders(4,5). IC is helpful for the evaluation of several conditions such as dry eye syndrome, cicatrizing conjunctivitis, vitamin A deficiency, limbal stem cell failure, effects of various medications, and ocular surface malignant tumors(6-13).
This study aimed to perform a descriptive analysis of the cellular alterations observed by IC of the ocular surface in patients with XP. The secondary objective was to assess the reliability of IC in diagnosing ocular surface squamous neoplasia (OSSN).
METHODS
This was a longitudinal study conducted from October 2018 to October 2019 to evaluate cellular changes in XP patients by performing IC for assessment of the ocular surface. Patients were recruited via social media (Facebook®, Instagram®) and messaging apps such as WhatsApp®. The study was approved by the Institutional Review Board of the CEP Universidade Federal de São Paulo - Unifesp, (approval number CAAE: 95105818.7.0000.5505). Written informed consent of patients was obtained prior to their enrolment.
Clinical assessment
The diagnosis of OSSN was based on slit-lamp examination and ancillary studies such as the assessment of the staining pattern of 1% toluidine blue (TB) eye drops. The TB staining patterns were categorized into 2 groups: "homogeneous" (score 1) and "stippled" (dot-shaped) (score A homogenous staining pattern was defined as the presence of homogeneous TB staining without stippled appearance). The distribution of dye uptake was then classified as follows: scattered (score 1); focal patches (score 2); and diffuse (score 3). Total scores ≤4 were considered "negative TB staining" and scores ≥5 were considered "positive TB staining." In case of any inter-observer disagreement regarding staining interpretation, the consensus opinion was selected as the final staining result. Additionally, TB-stained areas that also stained positive with fluorescein dye were not considered positive for TB staining(14) and anterior segment optical coherence tomography (OCT) pattern (severely thickened and hyperreflective epithelium with an abrupt transition between the normal and affected epithelium) was considered positive for OSSN(15).
IC: procedure
After administration of topical anesthesia with 0.5% deacaine hydrochloride (Anestalcon 0.5%, Alcon, São Paulo, Brazil), a strip of cellulose acetate filter paper with a diameter of 13 mm and pore size of 0.45 mm (Millipore HAWP01300, Bedford, US) was placed on the patient's ocular surface, pressed gently for 5 seconds, and then removed. The filter was immediately fixed for approximately 10 min in a solution containing glacial acetic acid, 37% formaldehyde, and ethyl alcohol in a volume ratio of 1:1:20. IC was performed on both eyes, when possible. In patients with a visible OSSN lesion, the filter paper was applied over the lesion. Otherwise, samples were collected from the nasal and temporal bulbar conjunctiva. Patients who underwent one-year follow-up examination were sampled at the same site as the baseline IC.
IC: interpretation
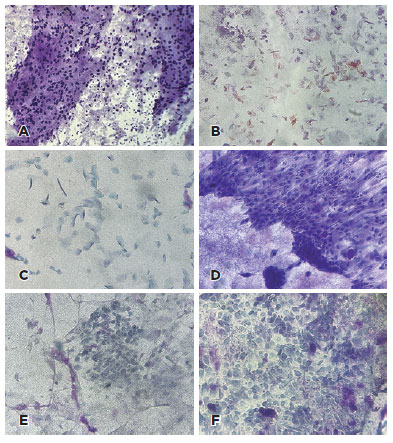
The presence or absence of goblet cells, inflammatory cells, keratinization; squamous metaplasia, and atypical epithelial cells was evaluated (Figure 1). Goblet cell densities were judged as present when Periodic Acid Schiff (PAS)-positive goblet cells containing mucin were identified in the sample and absent when no goblet cells were visible. Inflammatory cells were judged as present when neutrophils, or eosinophils, monocytes, macrophages, and lymphocytes could be identified. Keratinized cells were identified as enlarged, flattened cells with abundant organophilic (orange/yellow) or eosinophilic (pink) cytoplasm. There may be the presence of keratin filaments and pyknotic nuclei or even enucleated cells. Squamous metaplasia presents as a continuum of changes, including reduction/loss of goblet cells and gradual alterations of nongoblet epithelial cells (i.e., increased keratinization and stratification), as well as cellular enlargement and a decreased nuclear/cytoplasmic ratio(16). Atypical cells were identified by the presence of nuclear enlargement, hyperchromasia, irregular nuclear chromatin, increased NC ratio, and prominent nucleoli(12,17,18).
RESULTS
Clinical profile
Out of the 42 patients examined, IC was performed in 62 eyes of 34 participants (65% females). The mean age of participants was 29.6 ± 17 years (range, 7-62). At baseline examination, 15 of 62 eyes (24%) had a clinical diagnosis of OSSN. The baseline summary findings (Table 1) and detailed observations are tabulated (Table 2-Online supplement).
IC findings
The baseline IC showed goblet cells (47/62, 79%), inflammatory cells (12/62, 19%), keratinization (5/62, 8%), and squamous metaplasia (30/62, 48%). Representative IC findings are shown in figure 1.
Atypical cells: specificity/ sensitivity
Atypical cells were detected on IC in 18 patients (12 with OSSN and 6 without OSSN). Twelve eyes (19.3%) had atypical cells in IC and had clinical suspicion of OSSN, and three eyes (4.8%) had no atypical cells in IC and had clinical suspicion of OSSN. The detection of atypical cells in IC had 80% sensitivity, 87% specificity, 67% positive predictive value, and 93% negative predictive value for OSSN using clinical diagnosis as the reference standard (Table 3).

Correlation of IC findings with 1-year clinical course
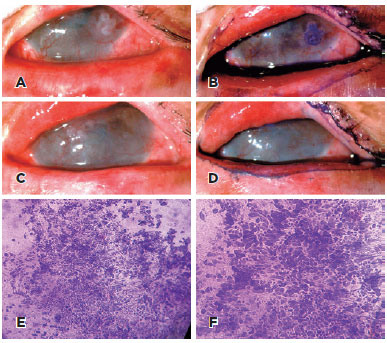
Thirteen participants completed one year of follow-up and repeated the IC examination. After one year, only one patient had residual OSSN despite treatment. The other patients experienced complete remission of OSSN after treatment. No recurrent or new tumors were observed. There were no cases with positive IC (presence of atypical cells) who later developed OSSN. At one-year follow-up, the IC findings were as follows: goblet cells (19/24, 75%), inflammatory cells (2/24, 8%), keratinization (3/24, 12%), and squamous metaplasia (13/24, 54%). The IC was false-positive for atypical cells in one patient (without OSSN) (Table 4). The patient (#23) had undergone successful treatment for OSSN with INF alpha-2b eye drops (1 million IU). At 1-year follow-up assessment, the patient showed no residual lesion on slit-lamp examination and TB staining despite the presence of atypical cells in IC (Figure 2).
DISCUSSION
Normal ocular surface epithelium, corneal and conjunctival, is a nonkeratinized multistratified plane consisting of 5-7 layers of polygonal cells. In the conjunctiva, numerous goblet cells also contribute to the tear film's formation(19). Diseases such as dry eye and ocular cicatricial pemphigoid are characterized by adaptive changes in the epithelium such as pathological transition to nonsecretory keratinized corneal and conjunctival cells, squamous metaplasia, and depletion of goblet cells(19-25). In our series of patients with XP, the detection of goblet cells in most eyes (47, 75%) is indicative of the high technical quality of the IC procedure. Approximately 50% (30 eyes) of our patients demonstrated squamous metaplasia, a reflection of the alterations in the surface epithelium secondary to XP.
Histopathology is the gold standard for the diagnosis of OSSN(26). However, only 50% of ophthalmologists perform biopsy to document a pathological diagnosis before starting topical therapy whereas others rely only on the clinical diagnosis possibly due to limited access to pathology services or costs(5,14,27). IC is a rapid and easy method and when performed by an expert pathologist, it has high accuracy for the initial diagnosis and follow-up of OSSN(5,12-14,17,28). IC is a minimally invasive technique that avoids surgical excision of the ocular surface, thereby sparing the corneal stem cells in the limbal area(12,14). Therefore, IC offers a safer alternative to repeated surgical biopsies(12). In the present study, pathologic confirmation of the diagnosis was not done to avoid further compromising the surface tissues in this high-risk population that has a tendency for development of new tumors or recurrences(12).
Management of OSSN can be divided into surgical and medical management. A follow-up study conducted in 2012 showed that surgery has remained the mainstay of therapy, but that there had been a significant increase in the use of topical therapy. With the advent of less invasive diagnostic modalities such as AS-OCT, confocal microscopy, and IC, there has been a shift toward less invasive management options. The three most commonly used topical treatments are interferon-α2b (IFN), 5-fluorouracil (5FU), and mitomycin-C (MMC). All three have shown similar outcomes in terms of tumor resolution rates and recurrence. The main differences relate to cost, storage, and side effect profile. Topical therapy avoids the risks associated with surgery and offers the benefit of treating the entire ocular surface. For larger tumors that extend onto the cornea, topical therapy avoids the risk of intraoperative corneal damage which may have significant visual consequences. Smaller tumors can be managed surgically or medically with equal success. Large tumors are preferentially managed by medical therapy to reduce surgical morbidity(29).
Although IC showed a moderate positive predictive value (67%) for diagnosis of OSSN in patients with XP, it had a high sensitivity (80%), specificity (87%), and negative predictive value (93%). In other words, IC that is negative for atypical cells can be informative in excluding OSSN.
It should be emphasized that the technique can be repeated, and patients can be followed until atypical cells are detected. It is important to note that IC is not a substitute for a possible biopsy for arriving at a definitive diagnosis and to guide appropriate treatment. In high-risk individuals, such as patients with XP, IC can be integrated into long-term management, as it may avoid unnecessary surgical biopsies.
ACKNOWLEDGMENTS
This study was supported by the Coordination for the Improvement of Higher Education Personnel (CAPES), Ministry of Education.
REFERENCES
1. Kleijer WJ, Laugel V, Berneburg M, Nardo T, Fawcett H, Gratchev A, et al. Incidence of DNA repair deficiency disorders in western Europe: xeroderma pigmentosum, Cockayne syndrome and trichothiodystrophy. DNA Repair (Amst). 2008;7(5):744-50.
2. DiGiovanna JJ, Kraemer KH. Shining a light on xeroderma pigmentosum. J Invest Dermatol. 2012;132(3 Pt 2):785-96.
3. Tang L, Cheng Y, Zhang Y, Ran D, Yang C, Gao M, et al. Novel compound heterozygous variants in the XPC gene identified in a Chinese xeroderma pigmentosum group C patient with ovarian teratoma. J Dermatol. 2018;45(11):e300-1.
4. Tananuvat N, Lertprasertsuk N, Mahanupap P, Noppanakeepong P. Role of impression cytology in diagnosis of ocular surface neoplasia. Cornea. 2008;27(3):269-74.
5. Barros JN, Lowen MS, Ballalai PL, Mascaro VL, Gomes JA, Martins MC. Predictive index to differentiate invasive squamous cell carcinoma from preinvasive ocular surface lesions by impression cytology. Br J Ophthalmol. 2009;93(2):209-14.
6. Tseng SC. Staging of conjunctival squamous metaplasia by impression cytology. Ophthalmology. 1985;92(6):728-33.
7. Nelson JD, Havener VR, Cameron JD. Cellulose acetate impressions of the ocular surface. Dry eye states. Arch Ophthalmol. 1983; 101(12):1869-72.
8. Fuchs GJ, Ausayakhun S, Ruckphaopunt S, Tansuhaj A, Suskind RM. Relationship between vitamin A deficiency, malnutrition, and conjunctival impression cytology. Am J Clin Nutr. 1994;60(2):293-8.
9. Puangsricharern V, Tseng SC. Cytologic evidence of corneal diseases with limbal stem cell deficiency. Ophthalmology. 1995; 102(10):1476-85.
10. Thiel MA, Bossart W, Bernauer W. Improved impression cytology techniques for the immunopathological diagnosis of superficial viral infections. Br J Ophthalmol. 1997;81(11):984-8.
11. Brandt JD, Wittpenn JR, Katz LJ, Steinmann WN, Spaeth GL. Conjunctival impression cytology in patients with glaucoma using long-term topical medication. Am J Ophthalmol. 1991; 112(3):297-301.
12. Tole DM, McKelvie PA, Daniell M. Reliability of impression cytology for the diagnosis of ocular surface squamous neoplasia employing the Biopore membrane. Br J Ophthalmol. 2001;85(2):154-8.
13. Barros JN, Almeida SR, Lowen MS, Cunha MC, Gomes JA. Impression cytology in the evaluation of ocular surface tumors: review article. Arq Bras Oftalmol. 2015;78(2):126-32.
14. Aliakbar Navahi R, Hosseini SB, Kanavi MR, Rakhshani N, Aghaei H, Kheiri B. Comparison of toluidine blue 1% staining patterns in cytopathologically confirmed ocular surface squamous neoplasias and in non-neoplastic lesions. Ocul Surf. 201917(3):578-83.
15. Kieval JZ, Karp CL, Abou Shousha M, Galor A, Hoffman RA, Dubovy SR, et al. Ultra-high resolution optical coherence tomography for differentiation of ocular surface squamous neoplasia and pterygia. Ophthalmology. 2012;119(3):481-6.
16. Calonge M, Diebold Y, Sáez V, Enríquez de Salamanca A, García-Vázquez C, Corrales RM, et al. Impression cytology of the ocular surface: a review. Exp Eye Res. 2004;78(3):457-72.
17. Nolan GR, Hirst LW, Wright RG, Bancroft BJ. Application of impression cytology to the diagnosis of conjunctival neoplasms. Diagn Cytopathol. 1994;11(3):246-9.
18. Solomon D, Davey D, Kurman R, Moriarty A, O'Connor D, Prey M, et al.; Bethesda 2001 Workshop. The 2001 Bethesda System: terminology for reporting results of cervical cytology. JAMA. 2002;287(16):2114-9.
19. Zuazo F, López-Ponce D, Salinas-Toro D, Valenzuela F, Sans-Puroja J, Srur M, et al. [Conjunctival impression cytology in patients with normal and impaired OSDI scores]. Arch Soc Esp Oftalmol. 2014; 89(10):391-6.
20. Murube J, Rivas L. Impression cytology on conjunctiva and cornea in dry eye patients establishes a correlation between squamous metaplasia and dry eye clinical severity. Eur J Ophthalmol. 2003;13(2):115-27.
21. Tseng SH, Chen YT, Huang FC, Jin YT. Seborrheic keratosis of conjunctiva simulating a malignant melanoma: an immunocytochemical study with impression cytology. Ophthalmology. 1999;106(8):1516-20.
22. Jalbert I, Madigan MC, Shao M, Ng J, Cheng J, Wong D, et al. Assessing the human lid margin epithelium using impression cytology. Acta Ophthalmol. 2012;90(7):e547-52.
23. Li S, Nikulina K, DeVoss J, Wu AJ, Strauss EC, Anderson MS, et al. Small proline-rich protein 1B (SPRR1B) is a biomarker for squamous metaplasia in dry eye disease. Invest Ophthalmol Vis Sci. 2008;49(1):34-41.
24. McNamara NA. Molecular mechanisms of keratinizing ocular surface disease. Optom Vis Sci. 2010;87(4):233-8.
25. Cvenkel B, Globocnik M. Conjunctival scrapings and impression cytology in chronic conjunctivitis. Correlation with microbiology. Eur J Ophthalmol. 1997;7(1):19-23.
26. Lee GA, Hirst LW. Ocular surface squamous neoplasia. Surv Ophthalmol. 1995;39(6):429-50.
27. Adesina A, Chumba D, Nelson AM, Orem J, Roberts DJ, Wabinga H, et al. Improvement of pathology in sub-Saharan Africa. Lancet Oncol. 2013;14(4):e152-7.
28. Sandeep Gupta RS. Namrata Sharma, Jeewan S Titiyal. ocular surface squamous neoplasia. Delhi J Ophthalmol. 2012;23(2):89-96.
29. Hӧllhumer R, Williams S, Michelow P. Ocular surface squamous neoplasia: management and outcomes. Eye (Lond). 2021; 35(6):1562-73.
Submitted for publication:
January 23, 2023.
Accepted for publication:
August 21, 2023.
Approved by the following research ethics committee: Universidade Federal de São Paulo – UNIFESP (CAAE: 95105818.7.0000.5505).
Disclosure of potential conflicts of interest: None of the authors have any potential conflicts of interest to disclose.
How to cite this article: