Arq. Bras. Oftalmol. 2026; 89 (3): 10.5935/0004-2749.2025-0392
Total: 496
Rodrigo Pessoa Cavalcanti Lira; Michelle Maria Teixeira Figueiredo Paiva; Gabriel Rocha Lira; Maria Isabel Lynch Gaete
DOI: 10.5935/0004-2749.2025-0392
ABSTRACT
PURPOSE: To evaluate the accuracy of a short-term intravitreal dexamethasone sodium phosphate challenge in predicting the anatomical response to a sustained-release dexamethasone implant (Ozurdex) in patients with refractory diabetic macular edema.
METHODS: This prospective, non-randomized, Phase 2 pilot study enrolled 12 pseudophakic eyes with diabetic macular edema refractory to anti-vascular endothelial growth factor (anti-VEGF) therapy. Participants underwent a challenge phase (Day 0) consisting of a single intravitreal injection of 0.08-mg dexamethasone sodium phosphate, followed by a maintenance phase (Day 7), during which all subjects received a 0.7-mg dexamethasone implant. The primary outcome was concordance between the anatomical response at Day 3 (post-dexamethasone sodium phosphate) and Day 60 (post-implant), defined as a ≥10% reduction in central macular thickness. Secondary outcomes included achieving a clinically "dry" macula (central macular thickness ≤300 μm) at Day 60, changes in best-corrected visual acuity, safety outcomes (intraocular pressure), and spectral-domain optical coherence tomography biomarker analysis.
RESULTS: The dexamethasone sodium phosphate challenge induced significant macular drying by Day 3 (median central macular thickness reduction, −21 μm; p=0.002). A positive response to dexamethasone sodium phosphate strongly predicted response to the dexamethasone implant, with a positive predictive value and specificity of 100%. The negative predictive
value was 80%. Irreversible biomarkers, including disorganization of retinal inner layers and ellipsoid zone disruption, were more prevalent among nonresponders (60% vs. 28.6%). Safety outcomes were acceptable; 16.7% of patients developed ocular hypertension, which was successfully managed with topical therapy.
CONCLUSION: A short-term dexamethasone sodium phosphate challenge is a safe,
low-cost, and highly specific predictor of dexamethasone implant efficacy. This "test-and-treat" strategy may optimize resource allocation in resource-constrained settings by identifying responders before high-cost implantation.
Keywords: Diabetic retinopathy; Macular edema; Dexamethasone; Drug implants; Biomarkers, pharmacological
INTRODUCTION
Diabetic macular edema (DME) remains the leading cause of vision loss among the working-age population worldwide, imposing a substantial burden on public health systems(1,2). The condition is characterized by breakdown of the blood-retinal barrier, resulting in extracellular fluid accumulation and structural disruption of the macula(1,3). Although vascular endothelial growth factor (VEGF) was historically identified as the primary driver of vascular permeability—leading to the widespread adoption of anti-VEGF agents as first-line therapy—real-world outcomes have demonstrated important limitations. Approximately 30%-50% of patients with DME show a suboptimal response to anti-VEGF monotherapy, presenting with persistent fluid and limited visual improvement despite intensive treatment(4-6).
Emerging evidence suggests that in chronic or refractory DME, disease pathophysiology shifts from a predominantly angiogenic, VEGF-driven process to one mediated by a broader inflammatory milieu, including cytokines such as interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α). This "cytokine switch" provides a strong biological rationale for corticosteroid therapy(7,8). The 0.7 mg biodegradable dexamethasone implant (Ozurdex; Allergan/AbbVie) targets this inflammatory cascade through sustained intraocular drug release, offering prolonged control of macular edema(9,10). However, widespread adoption of this implant—particularly in resource-constrained settings such as the Brazilian Unified Health System (SUS)—is limited by high cost and safety concerns, including cataract progression and intraocular pressure (IOP)elevation(11-13).
Clinicians, therefore, face a therapeutic dilemma. Withholding the implant due to cost or safety concerns may deprive true responders of meaningful visual recovery, whereas indiscriminate use leads to unnecessary financial burden and exposure to adverse effects in nonresponders—patients whose edema may be driven by irreversible structural damage or steroid-insensitive pathways. Consequently, there is an unmet need for a reliable biomarker to predict corticosteroid responsiveness.
In this context, we propose a novel diagnostic strategy using a low-cost, short-acting formulation of dexamethasone sodium phosphate (DSP) as a pharmacological "challenge test". DSP is highly soluble and has a short vitreous half-life (3.5-5.5 h), producing a rapid but transient therapeutic effect(14-17). We hypothesized that the early anatomical response to this corticosteroid pulse, assessed on Day 3, would reflect the presence of corticosteroid-sensitive pathways and thereby predict the efficacy of the sustained-release dexamethasone implant. This Phase 2 pilot study aimed to validate the predictive accuracy of this "test-and-treat" protocol in pseudophakic eyes with refractory DME.
METHODS
Study design and ethics
This prospective, non-randomized, interventional, longitudinal Phase 2 pilot study was conducted at a specialized retina center in Recife, Brazil. The study protocol adhered to the Declaration of Helsinki and the ICH-GCP guidelines. Ethical approval was obtained from the Ethics Committee of the Federal University of Pernambuco (CAAE: 80023924.5.0000.5208), and the trial was registered in the Brazilian Registry of Clinical Trials (REBEC: RBR-2bgcvwj). Written informed consent was obtained from all participants before enrollment.
Eligibility criteria
Patients with type 2 diabetes mellitus and center-involved DME refractory to anti-VEGF therapy were eligible for inclusion. Refractory DME was defined as a central macular thickness (CMT) >300 μm on spectral-domain optical coherence tomography (SD-OCT), with <10% reduction in CMT following at least six consecutive monthly intravitreal injections of 2 mg aflibercept. Inclusion was limited to pseudophakic eyes to eliminate cataract progression as a confounding factor for visual acuity outcomes. Best-corrected visual acuity (BCVA) was required to be between 1.3 logMAR (20/400 Snellen equivalent) and 0.2 logMAR (20/32 Snellen equivalent).
Exclusion criteria included recent treatment for DME (laser photocoagulation or intravitreal steroids within 4 months); panretinal photocoagulation within 6 months; intraocular surgery within 4 months; history of pars plana vitrectomy (PPV); open-angle glaucoma; steroid-induced ocular hypertension; or baseline IOP ≥21 mmHg.
Study protocol
The "challenge-and-treat" protocol consisted of two sequential phases:
1. Phase 1 (Challenge): On Day 0, participants received a single intravitreal injection of DSP (Decadron; Ache). A dose of 0.08 mg (0.04 mL of a 2-mg/mL solution) was administered via the pars plana under topical anesthesia using standard aseptic technique. Post-injection care included topical moxifloxacin 0.5%; no topical corticosteroids were prescribed.
2. Phase 2 (Implant): On Day 7, all participants received a 0.7 mg sustained-release dexamethasone implant (Ozurdex®; Allergan/AbbVie), irrespective of the response to the DSP challenge. This interval allowed for functional washout of DSP, corresponding to approximately five vitreous half-lives. The implant was administered via a shelved scleral injection technique.
Assessments and outcomes
Clinical evaluations included BCVA assessment using ETDRS charts (converted to logMAR), Goldmann applanation tonometry, and slit-lamp biomicroscopy. SD-OCT imaging (Cirrus; Zeiss) was performed at all study visits. Baseline OCT images were assessed for established prognostic biomarkers, including disorganization of retinal inner layers (DRIL), ellipsoid zone (EZ) disruption, hyperreflective foci, and cyst location.
The primary outcome was the predictive accuracy of the anatomical response at Day 3 (DSP effect) for the clinical response at Day 60 following dexamethasone implantation. A "DSP Responder" was defined as achieving a ≥10% reduction in CMT from baseline at Day 3. An "Ozurdex Responder" was defined as achieving either a ≥10% reduction in CMT from baseline or an absolute CMT ≤300 μm at Day 60. Secondary outcomes included mean changes in CMT and BCVA through Day 120 and the incidence of adverse events.
Statistical analysis
Sample size calculation indicated that a total of 12 participants would provide 80% power to detect a 0.5 standard deviation difference in macular thickness. Statistical analyses were performed using SPSS software (version 21.0; IBM Corp.). Data normality was assessed using the Shapiro-Wilk test. Longitudinal changes were analyzed using paired t tests or Wilcoxon signed-rank tests, as appropriate. Predictive performance was evaluated using 2 × 2 contingency tables to calculate sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV), with corresponding 95% confidence intervals. Changes in the frequency of categorical OCT biomarkers between Day 0 and Day 120 were analyzed using the McNemar exact test. Fishe's exact test was used to assess associations between categorical variables. Statistical significance was set at p<0.05. Due to the limited sample size, multivariate regression analysis was not performed to minimize the risk of model overfitting.
RESULTS
Twelve eyes from 12 participants completed the study protocol. Baseline characteristics were consistent with a population exhibiting advanced, chronic diabetic macular edema (Table 1).
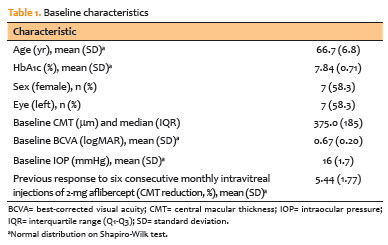
Anatomical and functional responses
The sequential corticosteroid regimen resulted in rapid and sustained anatomical improvement. Following the single 0.08 mg DSP injection, median CMT decreased significantly from 375.0 μm at baseline to 354.0 μm on Day 3 (p=0.002). After dexamethasone implant administration on Day 7, further macular drying was observed, with median CMT decreasing to 341.0 μm on Day 30 (p=0.002) and reaching a nadir of 305.0 μm on Day 60 (p=0.002). A mild trend toward recurrence was noted by Month 4, consistent with the known pharmacokinetics of the dexamethasone implant.
Functional outcomes were more modest compared with anatomical improvements. Mean BCVA improved from 0.67 logMAR at baseline to 0.57 logMAR at Day 60 (p=0.003), corresponding to a gain of approximately five ETDRS letters (Table 2).
Predictive accuracy
A reduction of ≥10% in CMT at Day 3 (DSP response) was a strong predictor of achieving a ≥10% reduction in CMT at Day 60 following dexamethasone implantation. The DSP challenge demonstrated a sensitivity of 88% (95% confidence interval [95% CI], 45-97) and a specificity of 100% (95% CI, 51-100). The positive predictive value (PPV) was 100% (95% CI, 65-100), indicating that all patients who responded to the low-cost DSP challenge subsequently responded to the sustained-release implant. The NPV was 80% (95% CI, 38-96; Table 3).
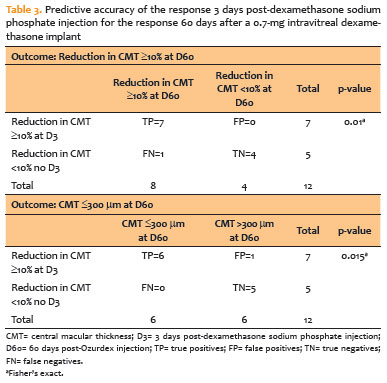
Similarly, a ≥10% reduction in CMT at Day 3 strongly predicted the achievement of a clinically "dry" macula (CMT ≤300 μm) at Day 60 (Ozurdex effect). In this analysis, sensitivity was 100% (95% CI, 60-100), specificity was 83.3% (95% CI, 45-97), PPV was 86% (95% CI, 54-97), and NPV was 100% (95% CI, 57-100; Table 3).
Safety
Mean IOP increased from 16.0 mmHg at baseline to 19.5 mmHg on Day 3 following DSP injection (p=0.005). After dexamethasone implant placement, IOP remained stable, with a mean of 18 mmHg at Day 60. Clinically significant ocular hypertension (>21 mmHg) occurred in two patients (16.7%) and was successfully controlled with topical timolol. No cases of endophthalmitis or retinal detachment were observed.
Biomarker analysis
SD-OCT analysis demonstrated a high prevalence of inflammatory and neurodegenerative features at baseline. The proportion of eyes with extrafoveal cysts decreased from 75% at baseline to 33.3% at Day 120 (p=0.063), while foveal cysts decreased from 58.3% to 16.7% over the same period (p=0.063), indicating a trend toward macular drying. Hyperreflective foci were present in all eyes at baseline and persisted at Day 120 despite the resolution of intraretinal fluid.
Markers of irreversible structural damage, including DRIL and EZ disruption, were present in 41.7% of eyes. These features were more frequently observed in nonresponders to the DSP challenge (60.0%) than in responders (28.6%); however, this difference did not reach statistical significance (p=0.558), likely due to the limited sample size.
DISCUSSION
This Phase 2 pilot study validates a pragmatic strategy for the management of refractory DME: the use of a low-cost pharmacological "challenge" to stratify patients before sustained-release corticosteroid therapy. Our findings demonstrate that a single intravitreal injection of DSP is a highly specific predictor of anatomical response to the 0.7 mg dexamethasone implant. With a PPV and specificity of 100%, the DSP challenge effectively functions as a "pharmacological biopsy", distinguishing eyes with corticosteroid-responsive edema from those with steroid-insensitive disease or irreversible structural damage.
The success of this strategy is supported by the complementary pharmacokinetic and pharmacodynamic profiles of the two corticosteroid formulations. DSP is a highly soluble ester that rapidly achieves high intravitreal peak concentrations, triggering an immediate genomic response that suppresses proinflammatory mediators such as IL-6 and VEGF. This short but potent "pulse" effect is sufficient to induce measurable intraretinal fluid reabsorption within 72 h(18,19). In contrast, the dexamethasone implant relies on a polylactic-co-glycolic acid matrix for sustained drug delivery and typically exhibits a pharmacological "lag phase" during the first postoperative week before stable therapeutic levels are achieved(20,21). A favorable response to the DSP challenge, therefore, indicates preserved glucocorticoid receptor signaling and functional fluid clearance pathways within the retina.
Spectral-domain OCT biomarker analysis revealed a high prevalence of inflammatory and neurodegenerative features, including DRIL and EZ disruption(22,23). Although significant anatomical improvement was achieved, functional gains were modest, averaging approximately five ETDRS letters. This dissociation highlights a key clinical reality in refractory DME: while anatomical drying is a necessary prerequisite for disease stabilization, it does not invariably translate into substantial visual recovery. The high prevalence of DRIL and EZ disruption in our cohort (41.7%) suggests that chronic neurodegenerative damage imposes a ceiling on functional improvement. In this context, preventing further vision loss and achieving a dry macula (CMT <300 μm) should be regarded as realistic and clinically meaningful therapeutic objectives.
The economic implications of this strategy are particularly relevant for public health systems such as the Brazilian SUS. The cost of a compounded DSP injection is minimal (less than $5), whereas the dexamethasone implant represents a substantial financial investment. Using the DSP challenge as a diagnostic "gatekeeper" enables early identification of nonresponders at negligible cost, thereby improving resource allocation. In addition, the challenge serves as a safety probe; identifying a clinically significant IOP response to a short-acting corticosteroid is inherently safer than detecting steroid responsiveness after implantation of a long-acting device.
Despite the excellent positive predictive performance, the NPV of 80% warrants careful interpretation. A negative DSP challenge does not categorically exclude benefit from the sustained-release implant, as a subset of patients without early response still demonstrated anatomical improvement at Day 60. This observation suggests that some eyes may require prolonged, steady-state corticosteroid exposure to overcome chronic inflammatory resistance—an effect not captured by the transient DSP pulse. Accordingly, a negative challenge should be interpreted as indicating reduced prognostic favorability rather than serving as an absolute contraindication to implantation.
The discrepancy between robust anatomical drying and modest visual improvement underscores the impact of chronic neurodegeneration in refractory DME. The high baseline prevalence of DRIL and EZ disruption likely established a functional "ceiling" for visual recovery. In this clinical context, preventing further vision loss and achieving a dry macula should be considered realistic and clinically meaningful therapeutic goals.
The primary limitation of this study is the small sample size (n=12), which is typical of a Phase 2 pilot investigation but results in wider confidence intervals. In addition, the absence of a control arm receiving dexamethasone implantation alone limits precise quantification of the incremental benefit conferred by the DSP induction challenge. The exclusion of phakic eyes further restricts generalizability; however, this design choice was necessary to isolate retinal function from lens-related confounding. Large, randomized controlled trials are therefore warranted to validate this treatment algorithm.
In conclusion, this study provides initial evidence that a short-term intravitreal challenge with DSP is a safe, accessible, and highly promising predictor of anatomical response to dexamethasone implantation in refractory DME. Based on these findings, we propose a clinical decision-making algorithm to optimize the management of refractory DME in resource-limited settings (Figure 1). This "test-and-treat" approach employs the DSP challenge as a diagnostic tool for both efficacy and safety before commitment to high-cost sustained-release therapy.
ACKNOWLEDGMENTS
This study was supported by an Undergraduate Research Scholarship from PIBIC-Propesqi (IC), UFPE, Grant PIA2924-2025 (Gabriel Rocha Lira).
The authors thank the clinical and nursing staff at the specialized retina center in Recife, Brazil, for their technical support and assistance with patient coordination. We are also grateful to the patients who participated in this research, whose contribution was essential for the completion of this Phase 2 pilot study.
AUTHORS' CONTRIBUTIONS
Significant contribution to conception and design: Rodrigo Pessoa Cavalcanti Lira, Michelle Maria Teixeira Figueiredo Paiva, Gabriel Rocha Lira, Maria Isabel Lynch Gaete. Data acquisition: Rodrigo Pessoa Cavalcanti Lira, Gabriel Rocha Lira. Data analysis and interpretation: Rodrigo Pessoa Cavalcanti Lira, Michelle Maria Teixeira Figueiredo Paiva, Gabriel Rocha Lira, Maria Isabel Lynch Gaete. Manuscript drafting: Rodrigo Pessoa Cavalcanti Lira, Michelle Maria Teixeira Figueiredo Paiva, Gabriel Rocha Lira, Maria Isabel Lynch Gaete. Significant intellectual content revision of the manuscript: Rodrigo Pessoa Cavalcanti Lira, Michelle Maria Teixeira Figueiredo Paiva, Gabriel Rocha Lira, Maria Isabel Lynch Gaete. Final approval of the submitted manuscript: Rodrigo Pessoa Cavalcanti Lira, Michelle Maria Teixeira Figueiredo Paiva, Gabriel Rocha Lira, Maria Isabel Lynch Gaete. Statistical analysis: Rodrigo Pessoa Cavalcanti Lira, Gabriel Rocha Lira. Obtaining funding: Gabriel Rocha Lira. Supervision of administrative, technical, or material support: Rodrigo Pessoa Cavalcanti Lira. Research group leadership: Rodrigo Pessoa Cavalcanti Lira.
REFERENCES
1. Tan GS, Cheung N, Simó R, Cheung GC, Wong TY. Diabetic macular oedema. Lancet Diabetes Endocrinol. 2017;5(2):143-55.
2. Teo ZL, Tham YC, Yu M, Chee ML, Rim TH, Cheung N, et al. Global prevalence of diabetic retinopathy and projection of burden through 2045: systematic review and meta-analysis. Ophthalmology. 2021; 128(11):1580-91.
3. Zhang J, Zhang J, Zhang C, Zhang J, Gu L, Luo D, et al. Diabetic macular edema: current understanding, molecular mechanisms and therapeutic implications. Cells. 2022;11(21):3362.
4. Madjedi K, Pereira A, Ballios BG, Arjmand P, Kertes PJ, Brent M, et al. Switching between anti-VEGF agents in the management of refractory diabetic macular edema: A systematic review. Surv Ophthalmol. 2022;67(5):1364-72.
5. Virgili G, Curran K, Lucenteforte E, Peto T, Parravano M. Anti-vascular endothelial growth factor for diabetic macular oedema: a network meta-analysis. Cochrane Database Syst Rev. 2023 Jun 27;2023(6):CD007419.
6. American Diabetes Association Professional Practice Committee. 12. Retinopathy, Neuropathy, and Foot Care: Standards of Care in Diabetes-2025. Diabetes Care. 2025;48(1 Suppl 1):S252-65.
7. Minaker SA, Mason RH, Lahaie Luna G, Farahvash A, Garg A, Bhambra N, et al. Changes in aqueous and vitreous inflammatory cytokine levels in diabetic macular oedema: a systematic review and meta-analysis. Acta Ophthalmol. 2022;100(1):e53-70.
8. Vujosevic S, Lupidi M, Donati S, Astarita C, Gallinaro V, Pilotto E. Role of inflammation in diabetic macular edema and neovascular age-related macular degeneration. Surv Ophthalmol. 2024; 69(6):870-81.
9. Park YG, Choi MY, Kwon JW. Factors associated with the duration of action of dexamethasone intravitreal implants in diabetic macular edema patients. Sci Rep. 2019;9(1):19588.
10. Iovino C, Mastropasqua R, Lupidi M, Bacherini D, Pellegrini M, Bernabei F, et al. Intravitreal dexamethasone implant as a sustained release drug delivery device for the treatment of ocular diseases: a comprehensive review of the literature. Pharmaceutics. 2020;12(8):703.
11. Roberti G, Oddone F, Agnifili L, Katsanos A, Michelessi M, Mastropasqua L, et al. Steroid-induced glaucoma: Epidemiology, pathophysiology, and clinical management. Surv Ophthalmol. 2020;65(4):458-72.
12. Liesenborghs I, Eijssen LM, Kutmon M, Gorgels TG, Evelo CT, Beckers HJ, et al. The molecular processes in the trabecular meshwork after exposure to corticosteroids and in corticosteroid-induced ocular hypertension. Invest Ophthalmol Vis Sci. 2020;61(4):24.
13. Jian YF, Zhang JS, Wan XH. Cataract Induced by Glucocorticoids. Clin Ophthalmol. 2025;19:3703-12.
14. Rodríguez Villanueva J, Rodríguez Villanueva L, Guzmán Navarro M. Pharmaceutical technology can turn a traditional drug, dexamethasone into a first-line ocular medicine. A global perspective and future trends. Int J Pharm. 2017;516(1-2):342-51.
15. Awwad S, Day RM, Khaw PT, Brocchini S, Fadda HM. Sustained release ophthalmic dexamethasone: in vitro in vivo correlations derived from the PK-Eye. Int J Pharm. 2017;522(1-2):119-27.
16. Fonseca AL, Panetta H, Nascimento MA, Lira RP, Arieta CE. Effect of intravitreal dexamethasone solution on the reduction of macular thickness in pseudophakic diabetic patients in a public hospital in Brazil: a randomized clinical trial. Clin Ophthalmol. 2019;13:1523-31.
17. Lira RP, Oliveira AY, Lira GR. Cost-effective treatment for diabetic macular edema using dexamethasone sodium phosphate. Arq Bras Oftalmol. 2025 ;88(1):e2024-0350.
18. Nehmé A, Edelman J. Dexamethasone inhibits high glucose-, TNF-α-, and IL-1β-induced secretion of inflammatory and angiogenic mediators from retinal microvascular pericytes. Invest Ophthalmol Vis Sci. 2008;49(5):2030-8.
19. Hui B, Yao X, Zhang L, Zhou Q. Dexamethasone sodium phosphate attenuates lipopolysaccharide-induced neuroinflammation in microglia BV2 cells. Naunyn Schmiedebergs Arch Pharmacol. 2020;393(9):1761-8.
20. Costello MA, Liu J, Chen B, Wang Y, Qin B, Xu X, et al. Drug release mechanisms of high-drug-load, melt-extruded dexamethasone intravitreal implants. Eur J Pharm Biopharm. 2023;187:46-56.
21. Costello MA, Liu J, Kuehster L, Wang Y, Qin B, Xu X, et al. Role of PLGA variability in controlled drug release from dexamethasone intravitreal implants. Mol Pharm. 2023;20(12):6330-44.
22. Luís ME, Sampaio F, Costa J, Cabral D, Teixeira C, Ferreira JT. Dril influences short-term visual outcome after intravitreal corticosteroid injection for refractory diabetic macular edema. Curr Eye Res. 2021;46(9):1378-86.
23. Nanji K, Grad J, Hatamnejad A, El-Sayes A, Mihalache A, Gemae M, et al. Baseline optical coherence tomography biomarkers associated with visual acuity in diabetic macular edema: a systematic review and meta-analysis. Ophthalmology. 2026;133(1):75-90.
Data Availability Statement: The datasets generated and/or analyzed during the current study are already available.
Edited by
Editor-in-Chief: Newton Kara-Júnior
Submitted for publication:
January 13, 2026.
Accepted for publication:
January 21, 2026.
Approved by the following research ethics committee: Universidade Federal de Pernambuco Campus Recife - UFPE/Recife (CAAE: 0023924.5.0000.5208).
Disclosure of potential conflicts of interest: The authors declare no potential conflicts of interest.
How to cite this article: