Arq. Bras. Oftalmol. 2026; 89 (3): 10.5935/0004-2749.2025-0248
Total: 205
Mateus Lins dos Santos1; Lucas Brito de Souza Florêncio2; Lucas Rocha Barreto de Almeida3; Lis Jacques Zwecker4; Melina Alves5; Ana Bastos de Carvalho6; Daniela Meirelles do Nascimento7; Luciana Bahia8; Beatriz D'Agord Schaan9,10; Gustavo Barreto Melo11; Fernando Korn Malerbi12
DOI: 10.5935/0004-2749.2025-0248
ABSTRACT
PURPOSE: This study aimed to identify barriers to diabetic retinopathy screening among a socioeconomically vulnerable urban population in northeast Brazil.
METHODS: A cross-sectional study was conducted during a diabetic retinopathy screening campaign at primary healthcare units. Ninety-five patients with diabetes underwent retinal examinations and completed a structured interview. Clinical, demographic, and socioeconomic data were collected.
RESULTS: The study population consisted predominantly of older adults (mean age: 60.7 ± 10.5 years), with a high prevalence of type 2 diabetes (99.0%) and low educational attainment. Most participants were economically inactive (81.1%) and reported low income (83.2%). Diabetic retinopathy and maculopathy were highly prevalent, affecting 50.0% and 22.9% of participants, respectively. Longer duration of diabetes was significantly associated with greater awareness of diabetic retinopathy (p=0.035), higher HbA1c levels (p<0.001), and increased prevalence of diabetic retinopathy (p=0.013) and maculopathy (p=0.002). Notably, 33.3% of participants reported difficulties attending medical appointments for diabetes management. In addition, 78.1% experienced challenges scheduling ophthalmologic evaluations, and 76.3% reported that no ophthalmologist was available in their city through the public healthcare system. Financial constraints also limited adherence to recommended dietary practices (90.4%) and impaired glycemic control, with more than half of participants reporting difficulty maintaining target glucose levels.
CONCLUSION: Major barriers to diabetic retinopathy screening included limited awareness of the importance of screening, financial hardship, and transportation challenges. Targeted educational initiatives and structural interventions such as expanded screening programs incorporating telemedicine and subsidized transportation—may improve screening adherence among vulnerable populations.
Keywords: Diabetic retinopathy; Mass screening; Health services accessibility; Health knowledge, attitudes, practices; Socioeconomic factors
INTRODUCTION
Diabetic retinopathy (DR) is the most common ocular complication of diabetes mellitus, affecting approximately 30%–40% of individuals with diabetes(1). The prevalence may reach up to 75% among individuals with more than 20 yr of disease duration(2). Globally, DR currently affects more than 100 million people, and this number is projected to increase to approximately 190 million by 2030(2,3). The prevalence of diabetic macular edema—a frequent complication of DR that requires ongoing treatment with costly anti-angiogenic agents to prevent vision loss—is also expected to rise, potentially affecting 24 million individuals by 2030(1).
Diabetic retinopathy often develops during the working years and remains the leading cause of visual impairment among the working-age population(1,2). The estimated direct medical cost of treating a patient with DR is approximately $40,825 over 5 yr(4), excluding indirect costs such as productivity loss.
Several factors are associated with the progression of DR, including poor glycemic control, systemic hypertension, longer duration of diabetes, dyslipidemia, and microalbuminuria(5). Screening programs facilitate early detection and timely intervention, including retinal photocoagulation and optimization of systemic disease management, thereby preventing severe visual complications(6).
However, numerous barriers hinder the implementation of effective DR screening programs, many of which are associated with socioeconomic conditions and the increasing prevalence of diabetes(7). These barriers include limited patient knowledge, negative attitudes toward screening, low motivation, shortages of healthcare professionals, insufficient training programs, limited retinal imaging infrastructure, and high healthcare costs(8-12). Such challenges are particularly pronounced in developing countries and in resource-limited regions of Brazil(13-17).
Previous studies conducted in Brazil have identified additional obstacles to DR screening, especially in underserved areas. Many individuals with diabetes have limited knowledge of the disease and its impact on ocular health and are often unaware of their right to specialized care within the public healthcare system(13,15). Furthermore, primary healthcare services exhibit significant structural inequalities across the country's macroregions, including inadequate professional training, lack of equipment, and limited effectiveness of health education initiatives(14,17). A substantial proportion of individuals with diabetes also remain undiagnosed, untreated, or inadequately controlled, contributing to the underdiagnosis and silent progression of DR(16). These factors underscore the need for integrated public health policies that address regional disparities and promote more effective strategies for screening and patient education in ocular health.
Given these challenges and the marked regional disparities in healthcare access for patients with diabetes, the present study aimed to evaluate the principal barriers to DR screening in a metropolitan region surrounding Aracaju, Sergipe, in northeastern Brazil.
METHODS
This cross-sectional observational study involved interviews with individuals diagnosed with diabetes mellitus. Participants were recruited consecutively from those who attended a DR screening activity and agreed to participate in the study. The study protocol was approved by the Ethics Committee of the Universidade Federal de Sergipe (CAAE: 64677723.0.0000.5546) and conducted in accordance with the principles of the Declaration of Helsinki. All participants were informed about the study objectives and procedures and provided written informed consent prior to participation.
Eligible participants included individuals of either sex aged 18 yr or older with a diagnosis of type 1 or type 2 diabetes mellitus and residing in the state of Sergipe, Brazil. Participants were excluded if they were unable to provide coherent responses during the interview, had incomplete questionnaire responses, or lacked point-of-care laboratory examination data. Data quality control procedures included verification of questionnaire completeness; participants with missing responses were excluded from subsequent analyses.
This study was conducted concurrently with a DR screening campaign organized by the Sergipe Society of Ophthalmology. Retinal examinations were offered free of charge at multiple primary healthcare units in São Cristóvão, Sergipe.
Participants underwent retinal imaging using the Eyer device (Phelcom Technologies, São Carlos, Brazil). Following imaging, participants completed a structured interview administered by a trained examiner. The interview questionnaire was adapted from Domingues et al.(18), which had previously been applied in a Portuguese-speaking population. The instrument assessed participants' knowledge, beliefs, and perceptions regarding social and economic aspects related to DR screening. The full questionnaire is provided as Questionnaire 1.
For Likert-scale questions, responses were converted into numerical scores ranging from 1 to 5, where 1 corresponded to "completely disagree" and 5 to "completely agree," enabling quantitative analysis of attitudes and perceptions.
Clinical parameters, including glycated hemoglobin (HbA1c), serum creatinine, and microalbuminuria, were obtained on-site during the screening using point-of-care devices: i-STAT 1 (Abbott, Chicago, Illinois) and Afinion 2 (Abbott, Chicago, Illinois). Information regarding insulin use was also recorded. These clinical data were collected immediately after completion of the questionnaire and integrated into the study dataset.
Fundus images were independently evaluated by two retinal specialists, who qualitatively assessed the presence and severity of DR and maculopathy. In cases of disagreement, a consensus decision was reached between the evaluators to ensure consistency in classification.
All data were recorded in electronic spreadsheets in XLSX format and analyzed using JASP statistical software (version 0.18.3.0). Descriptive analyses were performed for all variables. Continuous and ordinal variables were summarized using measures of central tendency (mean and median) and dispersion (standard deviation and range), whereas categorical variables were presented as frequencies and percentages.
For inferential analysis, continuous variables were compared using Student's t test, and categorical variables were analyzed using Pearson's chi-square test. Graphical representations were generated using the Matplotlib 3.9.1 library in Python 3.9.0. A p value <0.05 was considered statistically significant.
In addition to descriptive analyses, subgroup comparisons were conducted based on clinical and imaging findings. Participants were categorized according to the presence or absence of DR, maculopathy, and insulin use. These subgroup classifications were used to compare clinical parameters and questionnaire responses to identify potential associations.
RESULTS
Descriptive statistics
The clinical and sociodemographic characteristics of the study population are summarized in table 1. These data include demographic profile, comorbidities, medication use, and ophthalmologic examination findings. The frequency of responses to all questionnaire items is presented in table 2.

Among the main findings, 32 participants (33.6%) reported transportation-related difficulties that interfered with their ability to attend medical appointments. In addition, 77 participants (81.1%) reported difficulties scheduling ophthalmology appointments, and 73 (76.8%) indicated that no ophthalmologist was available in their city through the public healthcare system. These findings highlight significant structural barriers to accessing specialized eye care.
Socioeconomic factors also affected treatment adherence and self-care practices. A total of 88 participants (92.6%) reported difficulty maintaining an adequate diet because of financial constraints, and more than half stated that they were unable to maintain blood glucose levels within the recommended target range. Regarding awareness of screening, only 34 participants (35.8%) reported being informed about the purpose and procedure of DR screening.
Insulin use
Insulin-treated participants demonstrated significantly greater awareness of DR screening than non–insulin-treated participants. Specifically, 28 of 42 insulin-treated individuals (66.7%) reported awareness of DR screening, compared with 25 of 53 non–insulin-treated participants (47.2%; p=0.035). Similarly, insulin-treated participants showed greater understanding of the purpose and procedure of screening, with positive responses reported by 21 of 42 individuals (50.0%), compared with 13 of 53 non–insulin-treated participants (24.5%; p=0.015).
In addition, insulin-treated individuals more frequently reported having received recommendations to undergo DR screening (27/42; 64.3%) compared with non–insulin users (23/53; 43.4%; p=0.043). Insulin users also demonstrated higher mean agreement scores regarding their perceived future risk of developing DR (4.4 ± 1.0; range: 1–5) compared with non–insulin users (3.8 ± 1.3; range: 1–5; p=0.013).
Maculopathy
Participants with maculopathy showed slightly lower mean agreement scores on the belief that early identification of health problems is important (4.7 ± 0.7; range: 2–5) than those without maculopathy (4.9 ± 0.2; range: 4–5; p=0.039). A similar pattern was observed for perceptions regarding the importance of engaging in health-promoting activities (4.8 ± 0.4 vs 4.9 ± 0.1; p=0.011).
Awareness of DR was significantly associated with the presence of maculopathy. Among participants with maculopathy, 18 of 19 individuals (94.7%) reported knowledge of DR, compared with 19 of 64 individuals (29.7%) in the non-maculopathy group (p=0.013). Awareness of DR screening was also higher among individuals with maculopathy (15/19; 78.9%) than among those without maculopathy (32/64; 50.0%; p=0.025). Similarly, 17 of 19 participants with maculopathy (89.5%) reported receiving a recommendation to undergo DR screening, compared with 30 of 64 participants without maculopathy (46.9%; p=0.001). In addition, 13 of 19 individuals with maculopathy (68.4%) reported having received information about how DR screening is performed, compared with 19 of 64 participants without maculopathy (29.7%; p=0.002).
Diabetic retinopathy
Participants diagnosed with DR demonstrated significantly higher agreement scores regarding their perceived risk of developing retinopathy in the future (4.4 ± 0.8; range: 3–5) compared with those without DR (3.6 ± 1.4; range: 1–5; p=0.003).
Additional findings on screening awareness and socioeconomic factors
Awareness of DR screening was significantly associated with age. Participants who reported awareness had a lower mean age (58.2 ± 9.8 years; range: 39–82) compared with those who were unaware of screening (63.8 ± 10.7 years; range: 39–82; p=0.010).
Screening awareness was also associated with longer diabetes duration (12.5 ± 8.4 years; range: 1–35 ) compared with those unaware of screening (8.1 ± 6.4 years; range: 0.5–26; p=0.021), as illustrated in figure 1. In addition, individuals aware of screening had higher HbA1c levels (8.6 ± 2.3%) than those who were unaware (7.3 ± 1.9%; p=0.006).
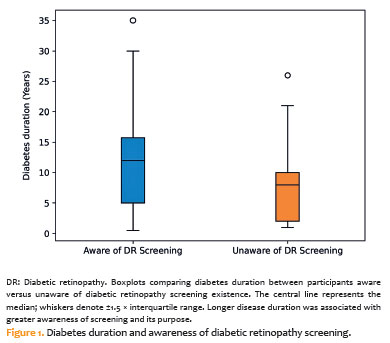
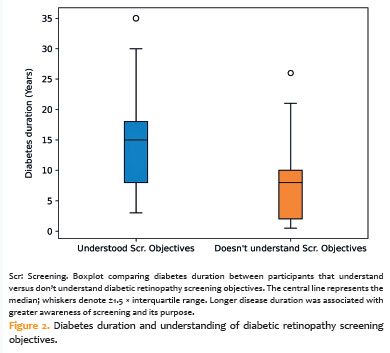
Understanding the purpose and procedure of DR screening was similarly associated with longer diabetes duration (14.1 ± 8.3 years vs 7.8 ± 6.2 years; p<0.001), as shown in figure 2, and with higher HbA1c levels (8.8 ± 2.3% vs 7.5 ± 2.1%; p=0.006).
Furthermore, participants who reported being able to maintain blood glucose levels within the recommended target range exhibited significantly lower HbA1c levels (7.3% ± 1.8%) than those who reported difficulties in glycemic control (8.7% ± 2.4%; p=0.003).
DISCUSSION
This study evaluated barriers to DR screening in a population predominantly composed of older adults with type 2 diabetes mellitus, low educational attainment, and limited income. Most participants were economically inactive and resided in urban areas. The high prevalence of DR (50.0%) and maculopathy (22.9%) observed in this study indicates a substantial burden of ocular complications among individuals with diabetes in this setting.
The literature on barriers to DR screening remains heterogeneous, with variations in study design and assessment methods(8–11). Commonly reported obstacles include excessive workload among healthcare providers(8,10), shortages of trained personnel(8,11,12), limited training opportunities(11,12), inadequate doctor–patient communication(10,19), limited patient availability(9), insufficient healthcare infrastructure(12), low health literacy(9,10,12,19), and the absence of visual symptoms during the early stages of the disease(10,19). Among these factors, lack of patient knowledge, poor perception of diabetes control, and the asymptomatic progression of DR were particularly consistent with the findings of the present study.
In our sample, individuals with a longer duration of diabetes demonstrated greater awareness of DR screening, consistent with previous reports(20,21). However, even among these participants, awareness did not necessarily translate into appropriate screening behavior, suggesting persistent misconceptions and limited health literacy. Kumar et al.(10) similarly reported that many patients delay ophthalmologic evaluation until visual impairment becomes evident. In Brazil, where a substantial proportion of individuals with diabetes have not completed secondary education(13,16), accessible communication strategies and culturally adapted educational materials are essential to improve patient engagement. Without such measures, preventive initiatives are unlikely to reach the most vulnerable populations, thereby limiting the potential impact of early detection programs.
Overall, knowledge regarding DR and its screening was limited across participant subgroups and was accompanied by low rates of previous ophthalmologic examinations and poor glycemic control. These findings are consistent with international evidence demonstrating low adherence to DR screening programs, even in high-income countries such as the United States(22,23) and Portugal(24), where organized regional screening programs do not achieve universal coverage. The similarity of these trends suggests that structural and educational barriers—rather than purely economic factors—are key determinants of low screening adherence. In Brazil, where population-based data remain limited, our findings likely reflect broader national challenges in implementing effective DR screening strategies(25).
Transportation difficulties, reported by approximately one-third of participants (Table 2), represented an important barrier to accessing healthcare services. Such challenges often arise from socioeconomic constraints, including advanced age and limited financial resources(9,26). Potential strategies to mitigate these barriers include providing free or subsidized transportation for high-risk patients, implementing home-based care models, and expanding teleophthalmology services(26,27). Financial hardship also negatively affected adherence to recommended dietary practices (Table 2), a relationship that has been widely documented in the literature(28).
Beyond individual-level barriers, systemic limitations within the healthcare infrastructure remain critical. Regional disparities in the distribution of ophthalmologists and ophthalmic imaging equipment hinder equitable access to DR screening services(14,17). Many primary healthcare units in underserved regions lack the infrastructure or referral networks required for timely ophthalmologic evaluation. These challenges reflect the structural limitations acknowledged in the Brazilian Clinical Protocol and Therapeutic Guidelines for Diabetic Retinopathy(29), which assign responsibility to state and municipal health authorities within the Unified Health System for organizing referral networks and ensuring regulated patient flow. Despite these formal policies, implementation remains inconsistent, often resulting in delayed diagnoses and preventable visual impairment. Strengthening coordination between primary and specialized care, investing in mobile retinal imaging units and teleophthalmology, and expanding professional training programs are essential strategies to improve the coverage and efficiency of DR screening nationwide.
Educational interventions also play a crucial role in improving screening uptake and glycemic control(27). The Brazilian Clinical Protocol and Therapeutic Guidelines for Diabetic Retinopathy highlight that underdiagnosis of diabetes and low population awareness represent major obstacles to early disease management. This underscores the importance of continuous health education integrated into primary care services. In this context, family physicians play a central role in coordinating diabetes management, facilitating referrals for ophthalmologic evaluation, and providing ongoing patient counseling(30).
Technological innovations, such as artificial intelligence–based screening systems, have demonstrated promising results in other settings. Dow et al.(31) reported a threefold increase in screening adherence and shorter intervals between screening and result reporting when artificial intelligence–assisted image analysis was implemented. However, these technologies remain at an early stage of adoption within the Brazilian Unified Health System and should therefore be considered future perspectives rather than immediate practical recommendations for the present context.
Several limitations of this study should be acknowledged. All data were self-reported and derived from a convenience sample, which may have introduced recall, interviewer, or selection bias. The cross-sectional design precludes causal inference, and the modest sample size limits the generalizability of the findings. Additionally, the lack of formal linguistic validation of the questionnaire may affect measurement reliability. The gender imbalance and predominance of urban participants may also have introduced selection bias. Furthermore, the analyses did not include adjustments for potential confounding variables or corrections for multiple statistical comparisons. Despite these limitations, this study provides valuable real-world evidence regarding barriers to DR screening in vulnerable populations and identifies critical factors that may inform strategies to improve the effectiveness of national screening initiatives.
In conclusion, this study conducted in a socioeconomically vulnerable urban population in northeastern Brazil identified significant barriers to DR screening. The study population—composed primarily of older adults with low educational attainment and limited income—demonstrated a high prevalence of DR and diabetic maculopathy. Key barriers included limited patient awareness and understanding of DR and the importance of screening, compounded by the asymptomatic nature of early disease stages. Socioeconomic factors, particularly financial constraints and transportation difficulties, also substantially limited access to screening services. These findings underscore the need for targeted interventions, including culturally adapted educational programs and structural improvements to healthcare services. Expanding screening availability through telemedicine, strengthening referral networks, and providing subsidized transportation may contribute to improving equitable access to ophthalmologic care among vulnerable populations.
ACKNOWLEDGMENTS
This study was supported by Fundo de Incentivo à Pesquisa e Eventos, Hospital de Clínicas de Porto Alegre (FIPE), Instituto de Avaliação de Tecnologia em Saúde (IATS), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS), and Sociedade Brasileira de Diabetes (SBD). Beatriz Schaan received research productivity grants from the CNPq Foundation, which also awarded a postdoctoral fellowship to Daniela Nascimento. Funding agencies do not have any role regarding the design of the study, data collection, analysis and interpretation of data, or writing the manuscript.
AUTHORS' CONTRIBUTIONS
Significant contribution to conception and design: Mateus Lins dos Santos, Beatriz Schaan, Daniela Nascimento, Luciana Bahia, Gustavo Barreto Melo, Fernando Korn Malerbi.
Data Acquisition: Mateus Lins dos Santos, Lucas Brito de Souza Florêncio, Lucas Rocha Barreto de Almeida, Lis Jacques Zwecker, Melina Vieira Alves.
Data Analysis and interpretation: Mateus Lins dos Santos, Ana Bastos de Carvalho, Beatriz Schaan, Daniela Nascimento, Luciana Bahia, Gustavo Barreto Melo, Fernando Korn Malerbi.
Manuscript Drafting: Mateus Lins dos Santos.
Significant intellectual content revision of the manuscript: Ana Bastos de Carvalho, Daniela Nascimento, Luciana Bahia, Beatriz Schaan, Gustavo Barreto Melo, Fernando Korn Malerbi.
Final approval of the submitted manuscript: Mateus Lins dos Santos, Lucas Brito de Souza Florêncio, Lucas Rocha Barreto de Almeida, Lis Jacques Zwecker, Melina Vieira Alves, Ana Bastos de Carvalho, Daniela Nascimento, Luciana Bahia, Beatriz Schaan, Gustavo Barreto Melo, Fernando Korn Malerbi.
Statistical analysis: Mateus Lins dos Santos.
Obtaining funding: Beatriz Schaan, Daniela Nascimento, Luciana Bahia.
Supervision of Administrative, technical, or material support: Mateus Lins dos Santos, Beatriz Schaan, Daniela Nascimento, Luciana Bahia, Gustavo Barreto Melo, Fernando Korn Malerbi.
Research group leadership: Fernando Korn Malerbi.
REFERENCES
1. Tan TE, Wong TY. Diabetic retinopathy: looking forward to 2030. Front Endocrinol (Lausanne). 2023 Jan;13:1077669.
2. Mozetic V, Daou JP, Martimbianco AL, Riera R. O que as revisões sistemáticas da Cochrane dizem sobre retinopatia diabética? Sao Paulo Med J. 2017;135(1):79-87.
3. Teo ZL, Tham YC, Yu M, Chee ML, Rim TH, Cheung N, et al. Global Prevalence of Diabetic Retinopathy and Projection of Burden through 2045: Systematic Review and Meta-analysis. Ophthalmology. 2021;128(11):1580–91.
4. Marques AP, Ramke J, Cairns J, Butt T, Zhang JH, Jones I, et al. The economics of vision impairment and its leading causes: A systematic review. EClinicalMedicine. 2022 ;46:101354.
5. Ghamdi AH. Clinical Predictors of Diabetic Retinopathy Progression; A Systematic Review. Curr Diabetes Rev. 2020;16(3):242–7.
6. Kashim RM, Newton P, Ojo O. Diabetic Retinopathy Screening: A Systematic Review on Patients' Non-Attendance. Int J Environ Res Public Health. 2018;15(1):157.
7. Gaskin DJ, Thorpe RJ Jr, McGinty EE, Bower K, Rohde C, Young JH, et al. Disparities in diabetes: the nexus of race, poverty, and place. Am J Public Health. 2014;104(11):2147–55.
8. Bastos de Carvalho A, Lee Ware S, Belcher T, Mehmeti F, Higgins EB, Sprang R, et al. Evaluation of multi-level barriers and facilitators in a large diabetic retinopathy screening program in federally qualified health centers: a qualitative study. Implement Sci Commun. 2021;2(1):54.
9. Prothero L, Cartwright M, Lorencatto F, Burr JM, Anderson J, Gardner P, et al.; EROS Study Investigators. Barriers and enablers to diabetic retinopathy screening: a cross-sectional survey of young adults with type 1 and type 2 diabetes in the UK. BMJ Open Diabetes Res Care. 2022;10(6):e002971.
10. Kumar S, Kumar G, Velu S, Pardhan S, Sivaprasad S, Ruamviboonsuk P, et al. Patient and provider perspectives on barriers to screening for diabetic retinopathy: an exploratory study from southern India. BMJ Open. 2020;10(12):e037277.
11. Watson MJ, McCluskey PJ, Grigg JR, Kanagasingam Y, Daire J, Estai M. Barriers and facilitators to diabetic retinopathy screening within Australian primary care. BMC Fam Pract. 2021;22(1):239.
12. Piyasena MM, Murthy GV, Yip JL, Gilbert C, Zuurmond M, Peto T, et al. Systematic review of barriers and enablers for access to diabetic retinopathy screening services in different income settings. PLoS One. 2019;14(4):e0198979.
13. Dias AF, Vieira MF, Rezende MP, Oshima A, Muller ME, Santos ME, et al. Perfil epidemiológico e nível de conhecimento de pacientes diabéticos sobre diabetes e retinopatia diabética. Arq Bras Oftalmol. 2010;73(5):414–8.
14. Lopes CG, Rocha TA, Thomaz ÉB, Tonello AS, Rocha NC, Duarte KM, et al. Desigualdades macrorregionais na atenção primária ao Diabetes Mellitus: comparação dos três ciclos do PMAQ-AB. Saude Debate. 2022;46(133):376–91.
15. Araújo RC, Costa PA, Celestino MN, Paixão ME, Santos CL, Andrade LD, et al. Conhecimento e utilização de direito à saúde por usuários com diabetes: pesquisa de métodos mistos. Esc Anna Nery. 2023;27:e20220298.
16. Tonaco LA, Velasquez-Melendez G, Moreira AD, Andrade FC, Malta DC, Felisbino-Mendes MS. Awareness of the diagnosis, treatment, and control of diabetes mellitus in Brazil. Rev Saude Publica. 2023;57:75.
17. Almeida TM, Souza MK, Miranda SS. Aspectos estruturais para a Diabetes Mellitus nas Unidades Básicas de Saúde em capitais brasileiras. Saude Debate. 2023;47(138):571–89.
18. Domingues JC. Barreiras à adesão ao rastreio da retinopatia diabética nos cuidados primários de saúde [dissertação]. Coimbra, Portugal: Universidade de Coimbra; 2018.
19. Fenwick EK, Aravindhan A, Tan NC, Tang WE, Ng LP, Wong WT, et al. Non-adherence to diabetes microvascular complications follow-up screening in the primary care population: Predictors, associated barriers, and facilitators. Diabetes Res Clin Pract. 2025;224:112193.
20. Borba AK, Arruda IK, Marques AP, Leal MC, Diniz AD. Knowledge and attitude about diabetes self-care of older adults in primary health care. Cien Saude Colet. 2019;24(1):125–36.
21. Rodrigues FF, Santos MA, Teixeira CR, Gonela JT, Zanetti ML. Relação entre conhecimento, atitude, escolaridade e tempo de doença em indivíduos com diabetes mellitus. Acta Paul Enferm. 2012;25(2):284–90.
22. Fathy C, Patel S, Sternberg P Jr, Kohanim S. Disparities in Adherence to Screening Guidelines for Diabetic Retinopathy in the United States: A Comprehensive Review and Guide for Future Directions. Semin Ophthalmol. 2016;31(4):364–77.
23. Liu Y, Zupan NJ, Shiyanbola OO, Swearingen R, Carlson JN, Jacobson NA, et al. Factors influencing patient adherence with diabetic eye screening in rural communities: A qualitative study. PLoS One. 2018;13(11):e0206742.
24. Pereira AM, da Silva Laureano RM, de Lima Neto FB. Five regions, five retinopathy screening programmes: a systematic review of how Portugal addresses the challenge. BMC Health Serv Res. 2021;21(1):756.
25. Chua J, Lim CX, Wong TY, Sabanayagam C. Diabetic Retinopathy in the Asia-Pacific. Asia Pac J Ophthalmol (Phila). 2018;7(1):3–16.
26. Scott Kruse C, Karem P, Shifflett K, Vegi L, Ravi K, Brooks M. Evaluating barriers to adopting telemedicine worldwide: A systematic review. J Telemed Telecare. 2018;24(1):4–12.
27. Moinul P, Barbosa J, Qian J, Chen ML, Mohaghegh M, Kaur H, et al. Does patient education improve compliance to routine diabetic retinopathy screening? J Telemed Telecare. 2020;26(3):161–73.
28. Nugawela MD, Gurudas S, Prevost AT, Mathur R, Robson J, Hanif W, et al. Ethnic disparities in the development of sight-threatening diabetic retinopathy in a UK multi-ethnic population with diabetes: an observational cohort study. J Pers Med. 2021;11(8):740.
29. Brasil. Ministério da Saúde. Clinical Protocol and Therapeutic Guidelines for Diabetic Retinopathy (PCDT–Retinopatia Diabética). Brasília (DF): Ministry of Health; 2021. Joint Ordinance No. 17, of December 20, 2021. Available from: https://www.gov.br/conitec/pt-br/midias/protocolos/20211220_portal_retinopatia_diabetica.pdf
30. Farford BA, Ahuja AS, Stewart MW, Naessens JM, Keith JJ. Screening for Diabetic Retinopathy with a Nonmydriatic Ultra-Wide-Field Retina Camera by Family Medicine Physicians. J Am Board Fam Med. 2021;34(1):231–7.
31. Dow ER, Chen KM, Zhao CS, Knapp AN, Phadke A, Weng K, et al. Artificial Intelligence Improves Patient Follow-Up in a Diabetic Retinopathy Screening Program. Clin Ophthalmol. 2023;17:3323–30.
Data Availability Statement: The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request, subject to justified conditions.
Edited by
Editor-in-Chief: Newton Kara-Júnior
Associate Editor: Carlos Augusto Moreira Neto
Submitted for publication:
August 26, 2025.
Accepted for publication:
February 10, 2026.
Approved by the following research ethics committee: Universidade Federal de Sergipe (CAAE: 64677723.0.0000.5546).
Disclosure of potential conflicts of interest: The authors declare no potential conflicts of interest.
How to cite this article: