Arq. Bras. Oftalmol. 2026; 89 (3): 10.5935/0004-2749.2024-0310
Total: 672
Laura Goldfarb Cyrino1; Lucas Macedo Nascimento2; Alexandre Yamada Fujimura Jr3; Vitor Expedito Alves Ribeiro4; Caio Ernesto do Rêgo Castro2; Dillan Cunha Amaral5; Jaime Guedes6; Lycia Maria M. P. Pedral Sampaio1
DOI: 10.5935/0004-2749.2024-0310
ABSTRACT
This study aimed to evaluate the efficacy and safety of topical losartan for treating corneal injuries and stromal fibrosis based on preclinical and clinical evidence. A systematic search was conducted in October 2024 following PRISMA guidelines across Embase, PubMed, Web of Science, and the Cochrane Library. Studies assessing topical losartan use in animal models or human patients with corneal injury were included. No randomized clinical trials were identified. Of 750 articles screened, 12 met the inclusion criteria – seven preclinical studies and five case reports. Preclinical evidence indicated that topical losartan at 0.2-0.8 mg/dL reduced stromal opacity and myofibroblast differentiation. Higher concentrations (8-80 mg/dL) offered no additional benefit and were associated with ocular surface irritation. The five case reports included 11 patients (12 eyes); eight eyes showed visual improvement, and no adverse effects were observed at a dose of 0.8 mg/dL. Topical losartan demonstrates potential as an antifibrotic agent for corneal injuries. However, variability in outcomes and dose-related toxicity at higher concentrations highlight the need for controlled clinical trials to confirm efficacy, establish optimal dosing, and ensure safety.
Keywords: Cornea; Epithelial cells; Myofibroblasts; Corneal opacity; Losartan
INTRODUCTION
Corneal pathology resulting from injury may occur secondary to traumatic, chemical, inflammatory, metabolic, autoimmune, infectious, surgical, or iatrogenic causes(1). Globally, this burden remains significant, with an estimated 1.5-2 million new cases of unilateral blindness each year(2). Corneal wound healing involves complex pathways often associated with persistent epithelial defects (PEDs), neurotrophic epitheliopathy, stromal fibrosis, corneal neovascularization (CNV), and endothelial damage, all of which can cause substantial visual impairment(3).
Damage to the epithelial basement membrane (EBM) and/or Descemet's basement membrane, coupled with delayed regeneration, allows continuous penetration of profibrotic cytokines such as transforming growth factor (TGF)-β1, TGF-β2, and platelet-derived growth factor (PDGF). These cytokines promote the differentiation of mature α-smooth muscle actin (SMA)+ myofibroblasts, which secrete large amounts of disorganized extracellular matrix components, leading to stromal scarring and loss of corneal transparency(4,5). Mesenchymal cells – including keratocytes, corneal fibroblasts, myofibroblasts, and bone marrow-derived fibrocytes – are the primary cellular contributors to stromal fibrosis(4,6). Myofibroblast development and fibrosis are primarily driven by TGF-β and other growth factors, including PDGF(5,7,8). Among these, the TGF-β isoforms are considered the most critical regulators of corneal wound healing.
Most opacity-inducing corneal injuries are treated surgically, typically through penetrating or lamellar keratoplasty(3). Pharmacological interventions have traditionally relied on topical corticosteroids, which are often insufficient in preventing corneal scarring or neovascularization and are associated with long-term adverse effects such as elevated intraocular pressure and cataract formation(2).
Recently, topical losartan has emerged as a potential therapeutic alternative. Losartan, an angiotensin II receptor antagonist, is widely used for hypertension, renal disease, and heart failure and is listed among the World Health Organization's Essential Medicines(9-12). Research suggests that losartan may reduce corneal scarring fibrosis by inhibiting the activation of extracellular signal-regulated kinase, thereby suppressing TGF-β-mediated signaling(3,5,13).
In preclinical models, losartan has been shown to decrease stromal haze and myofibroblast formation following alkali burn, simulated blast injury (irregular phototherapeutic keratectomy), acute incisions, and Descemet's membrane-endothelial excision(14-18). Clinical evidence remains limited and variable; only a few case reports have documented improvements in corneal fibrosis and visual acuity following topical losartan treatment(19,20).
Given the emerging yet heterogeneous evidence, this systematic review aims to synthesize the current preclinical and clinical data, distinguishing between these two bodies of evidence, to assess the efficacy and safety of topical losartan in the management of corneal injuries and stromal fibrosis.
METHODS
Literature search
A systematic review of the literature on topical losartan in the treatment of corneal diseases was conducted in accordance with the PRISMA guidelines(21). The search terms included ("losartan") AND ("eye" OR "corneal" OR "ocular" OR "cornea"). Searches were performed using PubMed, Embase, Web of Science, and the Cochrane Library databases.
Inclusion and exclusion criteria
Studies were included if they met the following criteria: investigated the effects of topical losartan on corneal diseases, were retrospective or prospective in design, and examined the effects of topical losartan in animals and humans participants.
Studies were excluded if only abstracts were available, the full text was inaccessible, or if they were review articles, letters, or commentaries.
Study selection and data extraction
The article selection process involved three steps. First, duplicate records were removed using Zotero software. Second, titles and abstracts were screened using Rayyan. Third, full-text articles were carefully reviewed for eligibility. Two independent reviewers performed the selection process, and discrepancies were resolved through discussion and consensus with senior investigators.
RESULTS
Study selection and baseline characteristics
As shown in figure 1, the database search identified a total of 750 articles: 96 from PubMed, 524 from Embase, 114 from Web of Science, and 16 from the Cochrane Database. After removing 182 duplicates and excluding ineligible studies, 22 articles remained for detailed evaluation based on the inclusion criteria. Subsequently, 10 articles were excluded according to the exclusion criteria, leaving 12 studies for the final analysis.
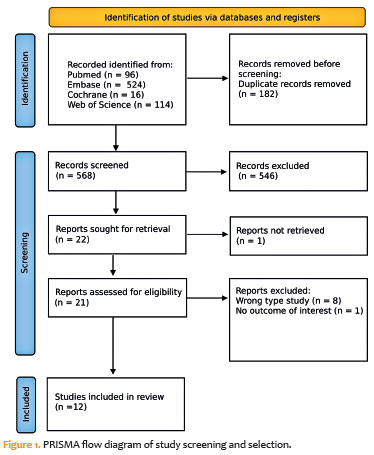
These comprised four case reports(13,19,20,22), one case series(23), and seven preclinical studies(14-18,24,25), which collectively analyzed 11 human patients (12 eyes) and 112 rabbits, of which 80 were assigned to the losartan treatment group. The characteristics of the human case studies are summarized in table 1, while details of the preclinical studies are presented in table 2. A comparative summary integrating clinical and preclinical findings – including model type, species, dosage, delivery method, outcomes, and limitations – is provided in table 3.
Case report studies
This review identified four case reports(13,19,20,22) and one case series(23).
Pereira-Souza et al.(19) reported a case involving a female patient who developed significant corneal fibrosis in the left eye following a complicated laser in situ keratomileusis (LASIK) procedure. The patient received topical losartan (0.8 mg/mL in balanced salt solution [BSS]), administered as 50 μL drops six times daily for 4.5 months. At the final follow-up, uncorrected distance visual acuity (UDVA) improved from 20/200 to 20/30, while corrected distance visual acuity (CDVA) improved from 20/30 to 20/25.
Rodgers et al.(20) described a male patient who developed corneal haze in the right eye after undergoing ultraviolet-A/riboflavin epithelium-off corneal cross-linking (CXL). The patient was treated with topical losartan (0.8 mg/mL, 50 μL in BSS, six times daily) for 3 months. At the final follow-up, CDVA returned to baseline. Corneal densitometry increased from 16.2 GSU preoperatively to 32.2 GSU at 80 days postoperatively, then improved to 27.9 GSU after three months of losartan therapy.
Wilson(13) reported a patient with corneal scarring following radial keratotomy who was treated with topical losartan (0.8 mg/mL). A marked reduction in corneal opacity was observed within 0.5 month, indicating a rapid therapeutic response.
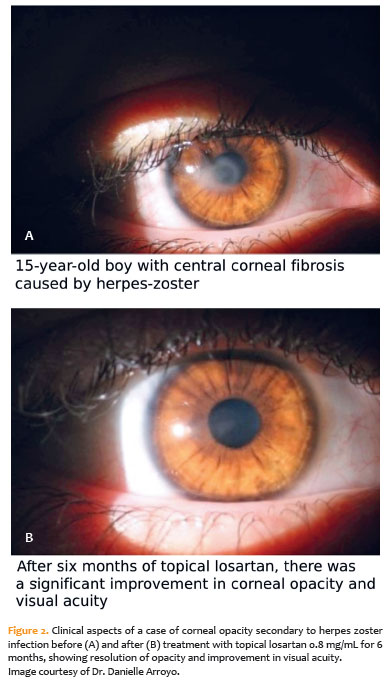
Santhiago et al.(22) described a 15-year-old male who developed corneal opacity in the right eye after an ocular manifestation of herpes zoster virus infection. Despite 1 year of treatment with oral valacyclovir and topical corticosteroids, no improvement in visual acuity or opacity was noted. After 6 months of topical losartan (0.8 mg/mL, six times daily), the opacity resolved, and visual acuity improved from 20/200 to 20/20. The images illustrating this case are shown in Figure 2A (before treatment) and Figure 2B (after treatment).
Finally, Burgos-Blasco and Moloney(23) reported a case series, including seven patients (eight eyes) with corneal fibrosis treated with topical losartan (0.8 mg/mL, four times daily for 6 months). The underlying causes included post-photorefractive keratectomy (PRK) haze, post-CXL haze, postadenoviral nummular keratitis, and post-PRK-CXL haze. At the final follow-up, best-corrected visual acuity (logMAR) improved from 0.28 ± 0.17 to 0.17 ± 0.11, although the change was not statistically significant (p=0.358). Among the treated eyes, five showed visual improvement, one remained unchanged, and two exhibited a one-line decrease in vision. Corneal densitometry and topographic parameters showed no significant differences (p>0.05) throughout the study. No systemic adverse effects were reported, and treatment tolerance was rated as very good to excellent by all patients.
Studies in rabbits
Seven preclinical studies were identified
In a preclinical study, Sampaio et al.(18) evaluated the therapeutic effects of topical and oral losartan on corneal stromal fibrosis in rabbits undergoing Descemetorhexis, a surgical procedure involving the removal of the central Descemet's membrane and corneal endothelium. The study specifically compared the efficacy of topical versus oral administration. Topical losartan (0.1 mg/mL) significantly reduced central stromal opacity, peripheral corneal scarring, and α-SMA-positive myofibroblast fibrosis compared with vehicle-treated eyes, indicating superior local efficacy. In contrast, oral losartan (5 mg/kg) did not significantly reduce fibrosis or stromal opacity, likely due to inadequate concentrations reaching the corneal stroma.
Further analysis of collagen type IV deposition – a fibrosis marker often upregulated by TGF-β – showed that topical losartan effectively decreased collagen type IV production in the posterior stroma, confirming its antifibrotic potential. Combined topical and oral administration did not produce additional benefits over topical treatment alone. Immunohistochemical analyses supported these findings, demonstrating markedly lower levels of α-SMA-positive myofibroblast markers in corneas treated with topical losartan. Additionally, reductions in collagen type IV and TGF-β1 levels in the posterior stroma highlighted the topical formulation's effective penetration and therapeutic action.
In a subsequent study, Sampaio et al.(15) evaluated the efficacy of 0.2 mg/mL losartan in BSS combined with 10 mg/mL prednisolone acetate in inhibiting corneal stromal myofibroblast formation and reducing fibrosis following alkali-burn injury. Alkali burns were induced using sodium hydroxide (NaOH) applied on filter paper for 1 min. The combination therapy significantly reduced both the area and intensity of corneal opacity, as determined by quantitative analysis of slit-lamp images processed using ImageJ software. Additionally, α-SMA staining – a marker of myofibroblast activity – was markedly decreased. Myofibroblasts were localized only to the posterior stroma at the end of follow-up, while the anterior and mid-stroma were repopulated with keratocan-positive keratocytes, suggesting an organized and favorable healing response.
Immunohistochemical evaluation revealed significantly lower collagen type IV staining intensity in the anterior stroma of corneas treated with losartan or combination therapy compared with controls. CNV occurred in all four eyes treated with BSS alone, two eyes treated with losartan, and one eye treated with prednisolone acetate. Notably, no eyes receiving the combination treatment developed neovascularization, demonstrating a protective effect against this common complication of corneal injury.
Martinez et al.(17) investigated whether deep central corneal incisions could close during topical losartan treatment and examined its effects on myofibroblast formation in rabbit corneas. Each rabbit received a 0.35 mm-deep radial incision extending from the corneal center to the limbus in one eye, approximated with a single 10-0 nylon suture 1 mm inside the limbus. The rabbits were treated with 50 µL of 0.8 mg/mL topical losartan in BSS or BSS (vehicle) six times daily for 1 month. Topical ciprofloxacin was applied until epithelial closure (within 5 days post-surgery). All incisions closed within 3-5 days without gaping or PEDs, and no corneal infections were noted. Although mean opacity levels were similar between the two groups, the losartan-treated eyes exhibited a significantly lower density of stromal myofibroblasts near the incision. The epithelial layer was thinner in the losartan group than in the vehicle group, though thicker than that in unwounded control corneas.
In another study, Sampaio et al.(14) evaluated the effect of topical losartan versus vehicle on myofibroblast formation and late haze development following PRK in rabbits. Each rabbit underwent −9.00 diopter PRK in one eye and was treated with 50 µL of 0.2 mg/mL losartan or vehicle six times daily for 1 month. Both groups also received topical ciprofloxacin three times daily until epithelial closure, but mitomycin C and corticosteroids were not used. The vehicle-treated group exhibited a mean opacity level of 93 pixels, while the losartan-treated group showed a significantly lower mean opacity of 77 pixels. Similarly, α-SMA staining was significantly reduced in the losartan-treated corneas (120 pixels) compared with vehicle (1,630 pixels), confirming losartan's antifibrotic effect.
Marked differences were observed in collagen type IV distribution. In vehicle-treated eyes, collagen type IV formed a broad band beneath the EBM, whereas in losartan-treated corneas, it was localized primarily to the basement membrane with minimal stromal deposition. Following PRK, TGF-β1 localization was intensified at the EBM in both groups, reflecting a reactive healing response. However, in the vehicle-treated corneas, a distinct stromal TGF-β1 band appeared below the collagen type I layer, indicating increased fibrotic signaling in untreated eyes.
Sampaio et al.(16) also examined the efficacy of topical losartan in reducing myofibroblast generation and corneal opacity following a blast injury-simulating irregular phototherapeutic keratectomy (PTK) in rabbits. Corneas were treated with 0.8 mg/mL losartan in BSS or vehicle six times daily for 6 weeks. Mean slit-lamp opacity did not differ significantly between groups. One eye in the losartan group (LOS-2) developed an epithelial defect at 4 weeks and was treated as an outlier. Excluding this case, topical losartan significantly reduced α-SMA intensity in the anterior stroma. The LOS-2 cornea, which developed an epithelial defect, showed high myofibroblast density, likely due to increased TGF-β penetration into the stroma. No significant differences were observed in keratocan or vimentin intensity, nor in TGF-β1 and collagen type IV levels between groups.
Dutra et al.(24) investigated the efficacy and safety of higher concentrations of topical losartan in a rabbit alkali-burn model. The 40 mg/mL dose caused severe ocular toxicity, leading to treatment discontinuation after 9 days. The 8 mg/mL dose produced PEDs and stromal opacity in five of six eyes after 1 month. In contrast, 0.8 mg/mL was well tolerated and significantly reduced corneal opacity in five of six corneas. Eyes treated with 8 mg/mL losartan exhibited higher α-SMA-positive cell counts (14.5 ± 2.8 vs. 3.5 ± 0.7, p=0.04) and greater vimentin expression (310 ± 64 vs. 132 ± 35, p=0.02) than those treated with 0.8 mg/mL, suggesting that higher concentrations may induce toxicity and diminish efficacy.
Finally, Dutra et al.(25) evaluated the safety and efficacy of topical losartan for treating established corneal fibrosis one month after alkali-burn injury in rabbits. Twenty-four rabbits received standardized alkali burns (0.75-N NaOH for 15 s) in one eye. After 1 month, the affected eyes were treated with topical losartan (0.8 mg/mL, 50 µL, six times daily) or vehicle (BSS). After 1 week and 1 month of treatment, topical losartan significantly reduced stromal myofibroblast density and increased stromal cell apoptosis, affecting both α-SMA-positive myofibroblasts and α-SMA-negative, vimentin-positive cells, compared with the vehicle group.
DISCUSSION
The findings of this systematic review provide compelling evidence supporting the efficacy of topical losartan in treating corneal injuries and preventing subsequent fibrosis in rabbits as well as suggestive evidence of its therapeutic potential in managing corneal haze in humans. The preclinical and clinical studies included in this review consistently demonstrate that topical losartan effectively reduces corneal haze, myofibroblast generation, and stromal fibrosis – key factors in the corneal healing process.
The four case reports(13,19,20,22) included in this review highlight the potential clinical benefits of topical losartan in human patients. Pereira-Souza et al.(19) and Rodgers et al.(20) reported substantial improvements in visual acuity and reductions in corneal haze following topical losartan administration in cases of corneal fibrosis after LASIK and corneal haze after collagen cross-linking, respectively. Wilson(13) described a patient with post-radial keratotomy scarring who demonstrated a rapid reduction in corneal opacity within only 0.5 month of treatment, suggesting early therapeutic efficacy. Similarly, Santhiago et al.(22) documented resolution of corneal opacity and restoration of visual acuity in a patient with herpes zoster-related corneal scarring after losartan therapy. Collectively, these reports underscore the translational potential of findings from animal models to human applications. Importantly, no adverse events were reported, indicating a favorable safety profile. However, given that the available human data are limited to small case series and isolated reports, these findings are inherently susceptible to publication bias, heterogeneity of models, and nonstandardized outcome measures. Although a formal risk-of-bias assessment is not applicable, these methodological limitations should be carefully considered when interpreting the results.
In contrast, the recent case series by Burgos-Blasco and Moloney(23) found that while some patients experienced improvements in visual acuity, the group-level change in best-corrected visual acuity was not statistically significant (p=0.358). Moreover, no significant improvements in corneal densitometry were observed. These findings suggest that losartan's therapeutic efficacy may vary among individuals, emphasizing the need for further clinical research to identify the patient subgroups most likely to benefit from treatment.
It is also essential to recognize clinical situations in which topical losartan may not provide a therapeutic advantage. For instance, transient haze after PRK typically resolves spontaneously without progressing to fibrosis, making antifibrotic therapy unnecessary. Similarly, temporary haze following riboflavin-ultraviolet corneal cross-linking is a physiological healing response that generally resolves without intervention. Because losartan primarily targets fibrotic pathways by inhibiting myofibroblast activity and reducing stromal opacity, its use in self-limiting conditions may be unwarranted. Further studies are needed to define the specific contexts in which losartan offers a clear clinical benefit versus those in which it may not be indicated(26-28).
The preclinical studies consistently confirmed the antifibrotic efficacy of topical losartan across various rabbit models of corneal injury. In a comprehensive study, Sampaio et al.(18) demonstrated that topical losartan significantly decreased central stromal opacity and α-SMA-positive myofibroblast fibrosis compared with oral losartan, underscoring the superior local efficacy of the topical formulation. Additionally, topical losartan reduced collagen type IV and TGF-β1 expression in the posterior stroma, confirming its role in inhibiting fibrotic signaling.
In another study, Sampaio et al.(15) showed that combining topical losartan with prednisolone acetate following alkali-burn injury markedly reduced corneal opacity, myofibroblast density, and neovascularization – a frequent complication of such injuries. These findings suggest a potential synergistic effect of losartan and corticosteroids, supporting their combined use in clinical management of corneal fibrosis secondary to chemical injury.
Martinez et al.(17) evaluated topical losartan's effects on corneal incision healing and found that treatment significantly reduced stromal myofibroblast density around surgical incisions, indicating enhanced wound healing and reduced fibrosis. Other studies in both animal and human models have shown that topical losartan creams or ointments can effectively minimize skin scar formation, further supporting its antifibrotic potential beyond ophthalmic use(29,30). Likewise, Sampaio et al.(14) confirmed that topical losartan reduced corneal opacity and myofibroblast presence after PRK, reinforcing its efficacy in preventing post-surgical corneal haze.
However, in a blast injury-simulating irregular phototherapeutic keratectomy (PTK) model, Sampaio et al.(16) found no significant differences between treatment and control groups in keratocan or vimentin expression, suggesting that losartan's benefits may depend on the severity or type of injury. The authors proposed that initial smoothing PTK combined with adjuvant topical losartan may better reduce stromal opacity after traumatic injuries with severe surface irregularities. Notably, in this model, 0.8 mg/mL losartan applied six times daily reduced anterior stromal α-SMA intensity compared with placebo, demonstrating partial efficacy.
An important observation from this study was the link between PEDs and increased myofibroblast differentiation. One outlier rabbit developed a PED and exhibited intense α-SMA expression, confirming earlier findings that delayed epithelial closure promotes early and excessive fibrosis(31,32). This reinforces the clinical importance of prompt epithelial wound closure to reduce both infection risk and fibrotic scarring.
Finally, the study by Dutra et al.(24), which explored higher concentrations of topical losartan, emphasized the importance of appropriate dosing. While 0.8 mg/mL was well tolerated and effective, higher concentrations such as 8 or 80 mg/mL induced ocular surface toxicity. The 40 mg/mL formulation caused severe discomfort and was discontinued after 9 days, while 8 mg/mL led to PEDs and worsened stromal opacity in most eyes. These findings highlight the narrow therapeutic window of losartan, where excessive dosing may result in adverse effects rather than improved outcomes(25).
Safety and adverse effects
The studies included in this review indicate that topical losartan is safe for use in corneal treatments. This favorable safety profile is an important factor supporting its potential for clinical application. However, preclinical studies using very high concentrations, such as 8 and 80 mg/mL, demonstrated that these doses were not only ineffective but also caused ocular irritation and surface toxicity. Therefore, maintaining an optimal therapeutic concentration is crucial to ensure both efficacy and safety.
Future applications
Although an increasing number of reports highlight the promising role of topical losartan in managing corneal fibrosis, further clinical investigations are needed to confirm its efficacy and safety in preventing and treating corneal scarring. Future studies could also evaluate its potential use after glaucoma surgery, particularly in reducing conjunctival bleb fibrosis and scarring(13).
This systematic review demonstrated that topical losartan exerts significant antifibrotic effects across several preclinical models and has shown potential benefits in isolated clinical cases. Nonetheless, the current evidence remains preliminary and heterogeneous. Thus, while topical losartan appears promising, its clinical application should still be regarded as experimental. Well-designed, randomized controlled trials are required to establish standardized dosing protocols, evaluate long-term safety, and confirm their therapeutic value before widespread clinical adoption.
ACKNOWLEDGMENTS
I would like to express my sincere appreciation to my coauthors for their valuable contributions and support, which have greatly enhanced the quality and rigor of this study. I am deeply grateful to Lycia Sampaio for her unwavering guidance and insightful expertise throughout the research process. Her extensive experience in this field and her thoughtful mentorship were invaluable to the success of this work.
AUTHORS' CONTRIBUTIONS
Significant contribution to conception and design: Laura Goldfarb Cyrino, Lucas Macedo Nascimento, Vitor Expedito Alves Ribeiro, Caio Ernesto do Rêgo Castro, Dillan Cunha Amaral, Alexandre Yamada Fujimura Júnior, Jaime Guedes, Lycia Maria Martins Pinho Pedral Sampaio.
Data acquisition: Laura Goldfarb Cyrino, Lucas Macedo Nascimento, Vitor Expedito Alves Ribeiro, Caio Ernesto do Rêgo Castro, Dillan Cunha Amaral, Alexandre Yamada Fujimura Júnior, Jaime Guedes, Lycia Maria Martins Pinho Pedral Sampaio.
Data analysis and interpretation: Laura Goldfarb Cyrino, Lucas Macedo Nascimento, Vitor Expedito Alves Ribeiro, Caio Ernesto do Rêgo Castro, Dillan Cunha Amaral, Alexandre Yamada Fujimura Júnior, Jaime Guedes, Lycia Maria Martins Pinho Pedral Sampaio.
Manuscript drafting: Laura Goldfarb Cyrino, Lucas Macedo Nascimento, Vitor Expedito Alves Ribeiro, Caio Ernesto do Rêgo Castro, Dillan Cunha Amaral, Alexandre Yamada Fujimura Júnior, Jaime Guedes, Lycia Maria Martins Pinho Pedral Sampaio.
Significant intellectual content revision of the manuscript: Laura Goldfarb Cyrino, Lucas Macedo Nascimento, Vitor Expedito Alves Ribeiro, Caio Ernesto do Rêgo Castro, Dillan Cunha Amaral, Alexandre Yamada Fujimura Júnior, Jaime Guedes, Lycia Maria Martins Pinho Pedral Sampaio.
Final approval of the submitted manuscript: Laura Goldfarb Cyrino, Lucas Macedo Nascimento, Vitor Expedito Alves Ribeiro, Caio Ernesto do Rêgo Castro, Dillan Cunha Amaral, Alexandre Yamada Fujimura Júnior, Jaime Guedes, Lycia Maria Martins Pinho Pedral Sampaio.
Statistical analysis: Laura Goldfarb Cyrino, Lucas Macedo Nascimento, Vitor Expedito Alves Ribeiro, Caio Ernesto do Rêgo Castro, Dillan Cunha Amaral, Alexandre Yamada Fujimura Júnior, Jaime Guedes, Lycia Maria Martins Pinho Pedral Sampaio.
Obtaining funding: not applicable.
Administrative, technical, or material support supervision: Laura Goldfarb Cyrino, Lucas Macedo Nascimento, Vitor Expedito Alves Ribeiro, Caio Ernesto do Rêgo Castro, Dillan Cunha Amaral, Alexandre Yamada Fujimura Júnior, Jaime Guedes, Lycia Maria Martins Pinho Pedral Sampaio.
Research group leadership: Laura Goldfarb Cyrino, Lucas Macedo Nascimento, Vitor Expedito Alves Ribeiro, Caio Ernesto do Rêgo Castro, Dillan Cunha Amaral, Alexandre Yamada Fujimura Júnior, Jaime Guedes, Lycia Maria Martins Pinho Pedral Sampaio.
REFERENCES
1. Barrientez B, Nicholas SE, Whelchel A, Sharif R, Hjortdal J, Karamichos D. Corneal injury: clinical and molecular aspects. Exp Eye Res. 2019;186:107709.
2. Dang DH, Riaz KM, Karamichos D. Treatment of non-infectious corneal injury: review of diagnostic agents, therapeutic medications, and future targets. Drugs. 2022;82(2):145-67.
3. Wilson SE. Magic Bullets: the coming age of meaningful pharmacological control of the corneal responses to injury and disease. J Ocul Pharmacol Ther. 2022;38(9):594-606.
4. Wilson SE. Corneal wound healing. Exp Eye Res. 2020;197:108089.
5. Wilson SE, Shiju TM, Sampaio LP, Hilgert GS. Corneal fibroblast collagen type IV negative feedback modulation of TGF beta: A fibrosis modulating system likely active in other organs. Matrix Biol. 2022;109:162-72.
6. Wilson SE. The Yin and Yang of mesenchymal cells in the corneal stromal fibrosis response to injury: the cornea as a model of fibrosis in other organs. Biomolecules. 2022;13(1):87.
7. Wilson SE. The corneal fibroblast: the Dr. Jekyll underappreciated overseer of the responses to stromal injury. Ocul Surf. 2023;29:53-62.
8. Wilson SE. Two-phase mechanism in the treatment of corneal stromal fibrosis with topical losartan. Exp Eye Res. 2024;242:109884.
9. Bui JD, Kimura B, Phillips MI. Losartan potassium, a nonpeptide antagonist of angiotensin II, chronically administered p.o. does not readily cross the blood-brain barrier. Eur J Pharmacol. 1992;219(1):147-51.
10. Carides GW. Losartan: a pharmacoeconomic review. J Med Econ. 2007; 10(4):573-85.
11. World Health Organization. World Health Organization Model List of Essential Medicines: 21st List 2019. Geneva: World Health Organization; 2019.
12. Te Riet L, van Esch JH, Roks AJ, van den Meiracker AH, Danser AH. Hypertension: renin-angiotensin-aldosterone system alterations. Circ Res. 2015;116(6):960-75.
13. Wilson SE. Topical Losartan: Practical Guidance for Clinical Trials in the Prevention and Treatment of Corneal Scarring Fibrosis and Other Eye Diseases and Disorders. J Ocul Pharmacol Ther. 2023;39(3):191-206.
14. Sampaio LP, Hilgert GS, Shiju TM, Santhiago MR, Wilson SE. Losartan Inhibition of Myofibroblast Generation and Late Haze (Scarring Fibrosis) After PRK in Rabbits. J Refract Surg. 2022;38(12):820-9.
15. Sampaio LP, Hilgert GS, Shiju TM, Santhiago MR, Wilson SE. Topical losartan and corticosteroid additively inhibit corneal stromal myofibroblast generation and scarring fibrosis after alkali burn injury. Transl Vis Sci Technol. 2022; 11(7):9.
16. Sampaio LP, Villabona-Martinez V, Shiju TM, Santhiago MR, Wilson SE. Topical losartan decreases myofibroblast generation but not corneal opacity after surface blast-simulating irregular PTK in rabbits. Transl Vis Sci Technol. 2023;12(9):20.
17. Martinez VV, Dutra BA, Sampaio LP, Shiju TM, Santhiago MR, Wilson SE. Topical Losartan inhibition of myofibroblast generation in rabbit corneas with acute incisions. Cornea. 2024;43(7):883-9.
18. Sampaio LP, Hilgert GS, Shiju TM, Murillo SE, Santhiago MR, Wilson SE. Topical losartan inhibits corneal scarring fibrosis and collagen type IV deposition after Descemet's membrane-endothelial excision in rabbits. Exp Eye Res. 2022;216:108940.
19. Pereira-Souza AL, Ambrósio R Jr, Bandeira F, Salomão MQ, Souza Lima A, Wilson SE. Topical Losartan for treating corneal fibrosis (Haze): first clinical experience. J Refract Surg. 2022;38(11):741-6.
20. Rodgers EG, Al-Mohtaseb Z, Chen AJ. Topical Losartan for Treating Corneal Haze After Ultraviolet-A/Riboflavin Collagen Cross-Linking. Cornea. 2024; 43(9):1165-70.
21. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
22. Santhiago MR, Sampaio LP, Arroyo D, Wilson SE, Moshirfar M, Del Risco N, et al. Addressing corneal opacity after herpes zoster infection. J Cataract Refract Surg. 2024;50(6):655-9.
23. Burgos-Blasco B, Moloney G. Topical losartan for corneal fibrosis: a case series with densitometry analysis. Cornea. 2024 Dec 13; https://doi.org/10.1097/ICO.0000000000003783.
24. Martinez VV, Dutra BA, Santhiago MR, Wilson SE. Effect of topical losartan in the treatment of established corneal fibrosis in rabbits. Transl Vis Sci Technol. 2024;13(8):22.
25. Dutra BA, Martinez VV, Santhiago MR, Wilson SE. Topical losartan dosage response and corneal toxicity at higher concentrations. Cornea. 2024; 44(5):637-45.
26. Wilson SE, Sampaio LP, Shiju TM, Hilgert GS, de Oliveira RC. Corneal opacity: cell biological determinants of the transition from transparency to transient haze to scarring fibrosis, and resolution, after injury. Invest Ophthalmol Vis Sci. 2022 Jan;63(1):22.
27. Katlun T, Wiegand W. [Haze und regression nach photorefraktiver Keratektomie (PRK) [Haze and regression after photoreactive keratectomy (PRK)]. Ophthalmologe. 2000;97(7):487-90. German.
28. Bulirsch LM, Weber C, Saßmannshausen M, Kohlhaas M, Holz FG, Loeffler KU, Herwig-Carl MC. [Histologische Veränderungen beim Keratokonus und Wundheilung nach kornealem Crosslinking [Histological changes in keratoconus and wound healing after corneal cross-linking]. Ophthalmologe. 2022 Apr;119(4):342-9. German.
29. Zheng B, Fang QQ, Wang XF, Shi BH, Zhao WY, Chen CY, et al. The effect of topical ramipril and losartan cream in inhibiting scar formation. Biomed Pharmacother. 2019;118:109394.
30. Khodaei B, Nasimi M, Nassireslami E, Seyedpour S, Rahmati J, Haddady Abianeh S, et al. Efficacy of topical losartan in management of mammoplasty and abdominoplasty scars: a randomized, double-blind clinical trial. Aesthetic Plast Surg. 2022;46(5):2580-7.
31. Sampaio LP, Martinez VV, Shiju TM, Hilgert GS, Santhiago MR, Wilson SE. Cell biology of spontaneous persistent epithelial defects after photorefractive keratectomy in rabbits. Transl Vis Sci Technol. 2023;12(5):15.
32. Wilson SE, Medeiros CS, Santhiago MR. Pathophysiology of corneal scarring in persistent epithelial defects after prk and other corneal injuries. J Refract Surg. 2018;34(1):59-64.
Data Availability Statement: The datasets generated and/or analyzed during the current study are already available.
Edited by
Editor-in-Chief: Newton Kara-Júnior
Associate Editor: Richard Y. Hida
Submitted for publication:
November 1, 2024.
Accepted for publication:
October 28, 2025.
Funding: This study received no specific financial support.
Disclosure of potential conflicts of interest: The authors declare no potential conflicts of interest.
How to cite this article: