INTRODUCTION
Chemical exposure to the eye can cause trauma ranging from mild irritation to the severe damage of ocular surfaces such as the conjunctiva, limbus, and anterior segment. Such damage has the potential to result in permanent vision loss. Chemical burns of the cornea cause superficial and deep neovascularization(1-3) which can lead to significant vision loss because of scar formation and lipid deposition(4). Regarding the impact of strong alkalis, ocular tissues have a limited ability to protect against such burns, which denature proteins and saponify lipids. Overall, chemical burns are responsible for an ocular trauma prevalence of between 7.7% and 18%.
Neovascularization is a poorly understood pathologic response of the cornea against chronic inflammation that is usually due to infection, sterile corneal ulceration, chemical or thermal injuries, or the immune rejection of corneal grafts. Following chemical burns, inflammatory cells such as polymorphonuclear leukocytes, and mesenchymal cells such as myofibroblasts, activated keratocytes, macrophages, and neovascularization factors are activated. Some of the factors responsible for the inducement of inflammation include vascular endothelial growth factor (VEGF), transforming growth factors, platelet-activating factor, basic fibroblast growth factor, and tumor necrosis factor-α(5-7). It has previously been reported that VEGF is upregulated in corneas with inflammation and vascularisation and that it is a significant angiogenic factor in corneal neovascularization(8,9).
Bevacizumab is a monoclonal antibody that binds to VEGF-A and all its isoforms. Its amino acid sequence comprises both human IgG (93%) and murine antibody (7%). It has been approved as an antiangiogenic pharmaceutical for the treatment of certain cancers and it has also been utilized to treat ocular neovascularization(10). In several studies it has been reported that topically or subconjunctivally applied bevacizumab results in the inhibition or regression of corneal neovascularizataion(11-13).
In this study we aimed to observe the effects of bevacizumab on experimentally produced corneal neovascularization in a rat model, and to compare the effects of this therapy when subconjunctivally or topically applied.
METHODS
Twenty-four male rats (Wistar-Albino) weighing between 250 g and 300 g and with two healthy eyes, were used in the study. All research obeyed the statement of the Association for Research in Vision and Ophthalmology (ARVO) for the Use of Animals in Ophthalmic and Vision Research. It was approved by the Local Ethics Committee for Animal Experiments of Kocaeli University. All rats were kept in individual cages and were managed under identical conditions.
Xylazine hydrochloride (5 mg/kg) (Rompun®, Bayer, Turkey) and ketamine hydrochloride (50 mg/kg) (Ketalar®, Eczacibasi, Turkey) were administered intraperitoneally for anesthesia and analgesia. For inducing corneal neovascularization, a previously described cauterization technique using silver nitrate was used(14). The right corneas of the rats were cauterized with a chemical applicator stick of 2 mm diameter, consisting of 75% silver nitrate and 25% potassium nitrate (Hemo Stop®, Istanbul, Turkey). This was touched onto the central corneas for eight seconds under an operating microscope. After cauterization, corneas and fornices were irrigated with 10 ml of normal saline in order to remove any residual silver nitrate. Burn stimulus responses were evaluated using the technique described by Mahoney and Waterbury(14). The burn stimulus response was scored as grade 0 (no blister), + 1 (small blister), + 2 (medium blister), or + 3 (large blister), according to lesions detected on the corneal surface (Figures 1, 2). Following cauterization, the burn stimulus scores were immediately calculated and treatment was begun.
The rats were categorized randomly into three groups of eight. The rats in the subconjunctival bevacizumab group were treated with a subconjunctival injection of 0.05 ml (1.25 mg) bevacizumab (Altuzan® 400 mg/16 ml, F. Hoffmann-La Roche Ltd., Basel, Switzerland). The rats in the topical group were treated topically with bevacizumab solution at a concentration of 10 mg/ml, and the rats in the control group received subconjunctival saline injections twice daily. The subconjunctival injections were performed with a 30 gauge needle under an operating microscope. Needles were inserted 2 mm posterior from the limbus at the superior temporal bulbar conjunctiva. Bevacizumab solution was prepared by dilution with normal saline under sterile situations and was kept at 4°C. The solution was applied twice per day (with the initial application performed immediately after cauterization) for seven days. All procedures were performed by the same investigator (O.O.).
On the eighth day, a slit-lamp microscopy examination was performed and digital photographs were taken under anesthesia and analgesia (Figure 3). For each eye, the neovascularization response was evaluated toward the centrally placed burn from the entire corneal circumference, as follows: Grade 0, no visible vessels in cornea; Grade 1.5, 1/4 distance to burn; Grade 2, 1/3 distance to burn; Grade 3, 1/2 distance to burn; Grade 4, 2/3 distance to burn; Grade 4.5, 3/4 distance to burn; and Grade 6, vessels reach burn(14). The area of corneal neovascularization was analyzed using a software program (Matlab(tm) R2007b version 7.5, Math Works, Natick, Massachusetts, USA). An investigator who was blinded to the study groups initially drew blood vessel borders and cornea circumferences on the photographs. The program then calculated the total cornea area and the neovascularized area. It then estimated the percentage of corneal neovascularization (neovascularized area/total cornea area × 100).
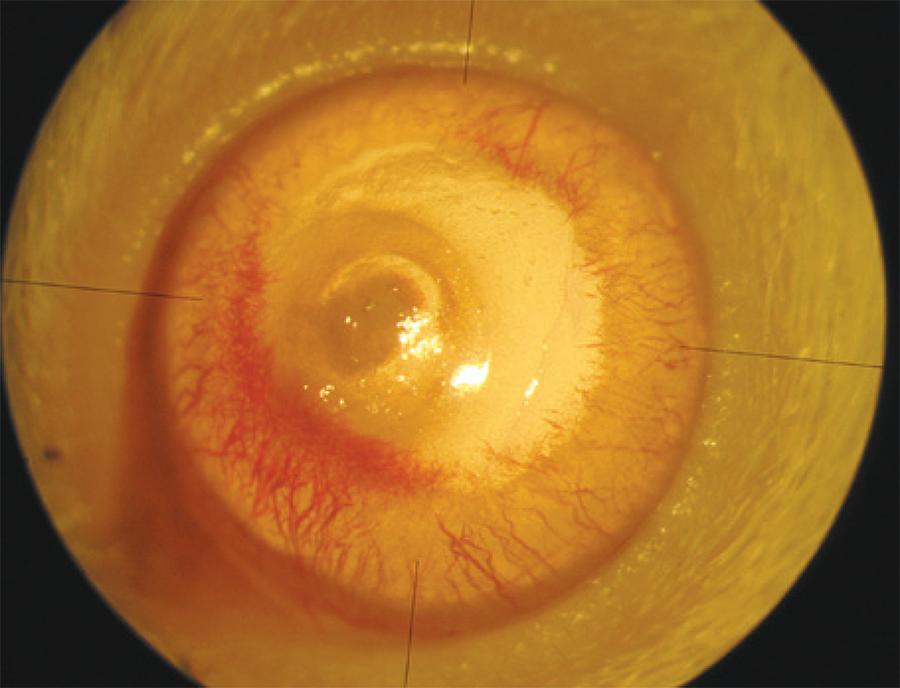
Figure 3 Image of an eye with corneal neovascularization after eight days of treatment. A scar formation can be observed on the central cornea. Neovascularization begins at the limbus and continues towards the burn area.
The rats were sacrificed on the eighth day using a high dose of pentothal sodium (Pentothal®, Abbott, Italy). The globes were then enucleated and fixed in 10% buffered formalin for 24 h. Corneas were then excised from the limbus and 5 µm thick sections were prepared. Sections were sliced from both the central region of the burn area and the intensive neovascularization area. They were then stained with haematoxylin-eosin and were analyzed using light microscopy. Sections were evaluated according to the intensity of neovascularization, the intensity of inflammation, and the fibroblast activity. Light microscopic inspections were performed on sections by an examiner (D.K.Y.) who was blinded to the study groups. The following scaling system, previously described in a study on corneal neovascularization, was used (Figure 4)(15).
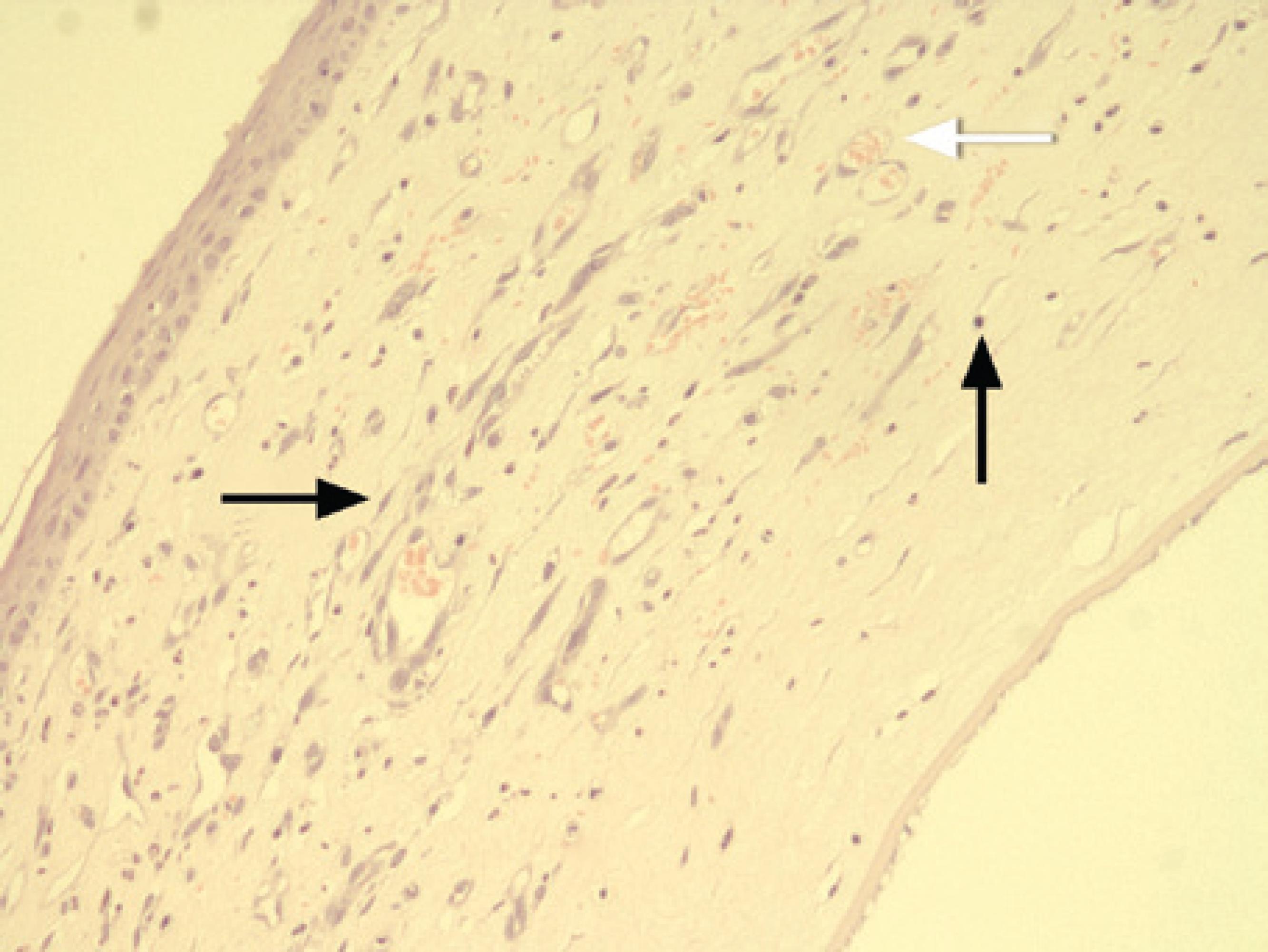
Figure 4 Histopathological preparation of a cornea. Inflammatory cells (thin arrow), fibroblasts (black thick arrow), blood vessels, and stained erythrocytes within the blood vessels (white thick arrow) can be seen. The corneal specimens were stained with hematoxylin-eosin and examined using light microscopy (magnification × 400).
Intensity of neovascularization: + 1: minimal or close to negative vascularization. + 2: limited or focal vascularization in the subepithelial and prestromal areas. + 3: cases intermediate to groups 2 and 4. + 4: diffuse and intense vascularization.
Intensity of inflammation: + 1: minimal or close to negative inflammation. + 2: focal, low count of mixed inflammatory cell types such as lymphocytes, neutrophil leukocytes, and eosinophil leukocytes). + 3: cases intermediate to groups 2 and 4. + 4: intense, diffuse, mixed inflammatory cell types.
Fibroblast activity: + 1: minimal or close to negative fibroblast activity. + 2: focal fibroblast activity. + 3: cases intermediate to groups 2 and 4. + 4: diffuse and intense fibroblast activity.
Statistical analysis
Statistical analyses were performed using SPSS 16.0 (SPSS(tm) Inc., Chicago, IL, USA). Data were evaluated as the mean ± standard deviation (SD). The Kruskal-Wallis test was performed for multiple comparisons of groups. When a statistically significant difference was found, a pair-wise comparison of groups was performed using the Mann-Whitney U test. Significance was defined as a p value <0.05.
RESULTS
The mean burn stimulus score was 2.0 ± 0.7 for the control group, 1.8 ± 0.6 for the subconjunctival bevacizumab group, and 1.7 ± 0.4 for the topical bevacizumab group. No statistical differences were found between the groups (p=0.730).
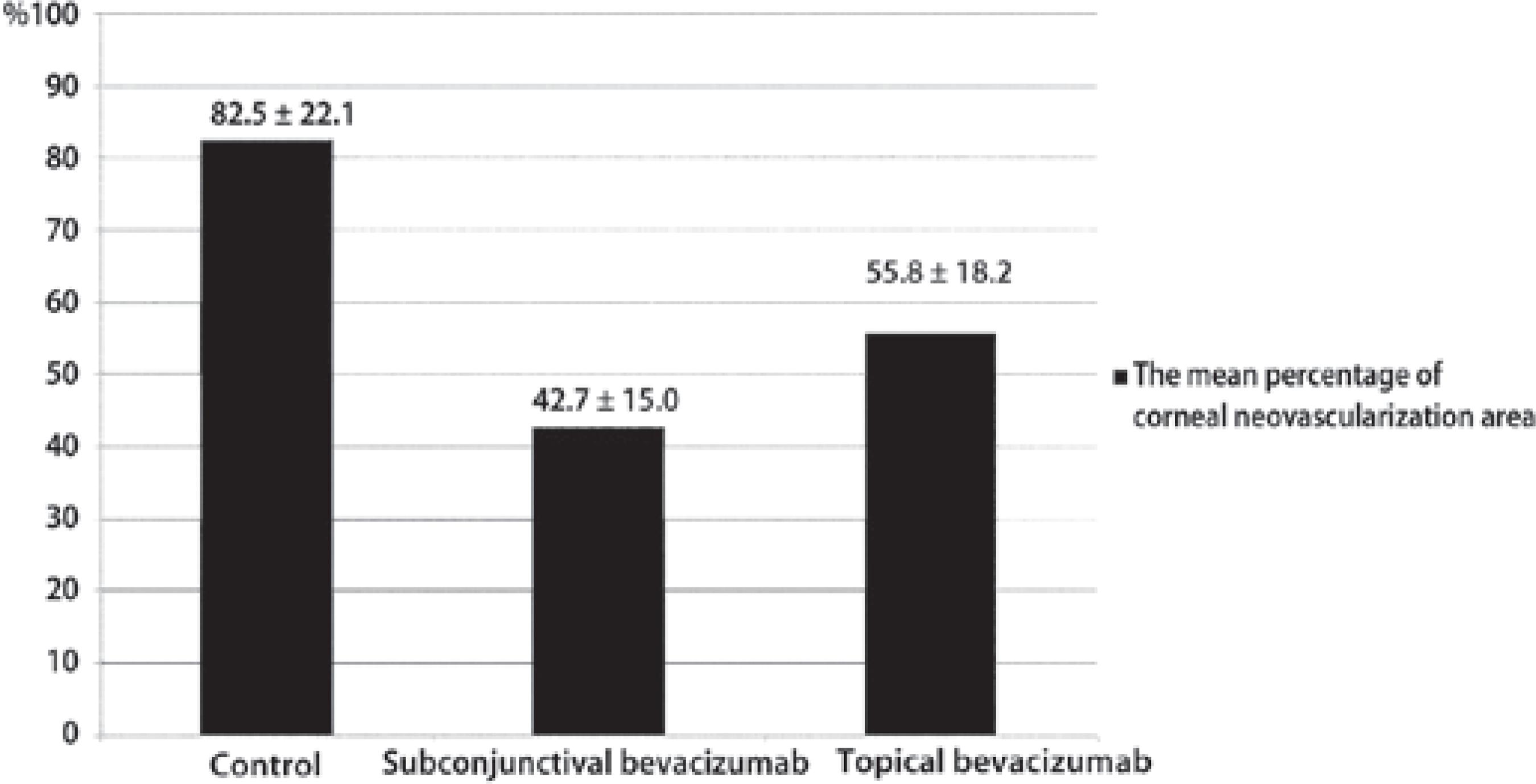
The mean neovascularization score was 5.5 ± 0.8 for the control group, 2.8 ± 1.7 for the subconjunctival bevacizumab group, and 3.5 ± 1.6 for the topical bevacizumab group. The mean neovascularization scores of the subconjunctival and topical bevacizumab groups were statistically lower than the control group (p=0.015 and p=0.035, respectively). In a comparison between the subconjunctival and topical bevacizumab groups, the mean neovascularization score was lower in the subconjunctival group. However, the difference was not statistically significant (p=0.512).
The mean percentage of corneal neovascularization area is shown in figure 5. It was found to differ significantly between groups (p=0.001). Both the subconjunctival and topical bevacizumab treatments inhibited corneal neovascularization. However, there was no statistically significant difference between these two treatments (p=0.142).
In the histopathological evaluation, both treatment groups displayed less neovascularization, inflammation, and fibroblast activity than the control group (p<0.05) (Table 1). The difference between the subconjunctival and topical bevacizumab groups was not statistically significant for the following evaluations: intensity of neovascularization (p=0.298), intensity of inflammation (p=0.960), and fibroblast activity (p=0.225).
Table 1 The mean (± SD) results for the histopathological evaluations of the three groups
| Groups | Intensity of neovascularization | Intensity of inflammation | Fibroblast activity |
|---|---|---|---|
| Control | 3.5 ± 0.5* | 3.5 ± 0.5* | 3.3 ± 0.7* |
| Subconjunctival bevacizumab | 2.4 ± 0.5 | 2.2 ± 0.7 | 2.1 ± 0.6 |
| Topical bevacizumab | 2.7 ± 0.4 | 2.8 ± 0.3 | 2.5 ± 0.5 |
| P a | 0.07* | 0.014* | 0.015* |
a= Kruskal-Wallis test
*= indicates a statistically significant difference compared to the other groups.
DISCUSSION
Corneal neovascularization originates from venules and capillaries of the pericorneal (limbal) plexus. Types of corneal neovascularization include vascular pannus, superficial vascularization, and stromal vascularization. Many medical and surgical treatments have been reported for the management of corneal neovascularization. Traditionally, topical steroids are recommended, primarily due to their suppression of actively growing neovascularization. Medical treatments may also include non-steroidal anti-inflammatory agents such as rapamycin, cyclosporine A, gene therapy approaches, methotrexate, and anti-VEGF agents. Laser and surgical treatments include the use of argon lasers, yellow lasers, Nd:YAG lasers, superficial keratectomy, diathermy, cauterization, and photodynamic therapy(16).
VEGF is a family of proteins comprising VEGF-A, -B, -C, and -D, the viral VEGF homologue VEGF-E, and placental growth factor(10). VEGF-A is one of the most important mediators of angiogenesis. It is upregulated under conditions of neovascularization and it plays an important role in the development of pathological angiogenesis in inflammatory, neoplastic, and vascular diseases of the eye. Numerous studies have shown that anti-VEGF agents, used either alone or in combination with steroids, verteporfin, or anti-tumor necrosis factor-α microantibody, are effective in the treatment of corneal neovascularization(17). Anti-VEGF agents reported for the treatment of corneal neovascularization include bevacizumab, pegaptanib, and ranibizumab. It has been reported that bevacizumab, ranibizumab, pegaptanib, and trastuzumab all have the ability to inhibit corneal neovascularization, and that bevacizumab is the most effective treatment in an experimental rat model(18). Various experimental and clinical studies have reported that bevacizumab inhibits corneal neovascularization when used at different doses both topically and subconjunctivally(11-13).
In our comparison of topical and subconjunctival applications of bevacizumab, there were no statistical differences regarding the neovascularization score, the corneal neovascularization area, or the histopathological evaluation. Compared to the control group, we found that corneal neovascularization, inflammation, and fibroblast activity decreased in both the topical and subconjunctival bevacizumab treatment groups. These results indicate that both treatment methods inhibit the development of corneal neovascularization. Furthermore, we have demonstrated that both treatments produce meaningful anti-inflammatory effects and reduce fibroblast activity. However, upon careful examination of the data we propose that the subconjunctival application appears to be more effective, although we accept that the difference is not supported statistically. Firstly, this difference in effect may be related to the limited penetration of topical bevacizumab through the corneal epithelium and/or its rapid clearance by tears(19,20). Secondly, the twice daily application of topical bevacizumab could be an insufficient treatment dose in this study design.
There have been several previously published studies comparing topical and subconjunctival bevacizumab treatments(21-23). Using rat corneas cauterized with silver nitrate sticks, Öner et al.(21) injected 0.05 ml (1.25 mg) of bevacizumab subconjunctivally on the first, fourth, and seventh days, or (in a second group) applied 4 or 12.5 mg/ml bevacizumab topically twice daily. They reported that both treatments were safe and effective in controlling corneal neovascularization. This result is supported by another finding demonstrating that both subconjunctival (1, 5, and 25 mg/ml) and topical (25 mg/ml) bevacizumab treatments prevent rat corneas from neovascularization(22). However, Dastjerdi et al.(23), in a model of vascularized corneal transplantation, investigated corneal graft survival rates following their treatment with either topical or subconjunctival bevacizumab. They showed that the regression of corneal neovascularization was more profound when subconjunctival treatment was applied. Furthermore, Ahmed et al.(19) compared topical (12.5 mg/ml, three times daily) and subconjunctival (5 mg and 10 mg) bevacizumab treatments in an experimental rabbit model of corneal neovascularization, and found a significant decrease in the amount of neovascularization, particularly in the subconjunctival group. They reported that the subconjunctival treatment was superior because of the rapid clearance of the topically applied drug by tears and its subsequent low concentration on the ocular surface. It was suggested that topically applied bevacizumab has a limited capacity to access corneas with an intact epithelium and that, contrastingly, subconjunctivally injected bevacizumab penetrates the corneal stroma in eyes with an intact corneal epithelium(19). Conversely, Kim et al. reported that topical administration of bevacizumab produced a longer lasting anti-angiogenic effect than subconjunctival injection in corneal neovascularization following chemical injury in rats. They suggested that the half-life of bevacizumab after subconjunctival injection was probably not sufficiently long to demonstrate a continuous effect(24).
In this investigation we replicated the methods of Mahoney and Waterbury(14), by touching an applicator stick made from silver nitrate onto the cornea in order to induce neovascularization, and by conducting the study over seven days duration. In experimental studies of corneal neovascularization there are some differences of opinion regarding the timing of the anti-antigenic treatment and the duration of the investigation. The duration of previously reported studies has ranged from one to three weeks. For example, McCulley(25) grouped the clinical progress of chemical injuries into four distinct phases: immediate, acute (0-7 days), early reparative (7-21 days), and late reparative (>21 days). Regarding treatment, the injured eye or eyes are immediately irrigated until the pH returns to neutral, after which medical treatment is rapidly begun in order to promote epithelial wound healing, minimize inflammation, minimize ulceration, and control intraocular pressure(26). In the study of Mahoney and Waterbury(14), neovascularization appeared on the third day, reached a maximum level on the fifth post cauterization day, and decreased after 7-10 days. Other studies have shown that bevacizumab significantly inhibits neovascularization when injected simultaneously with the chemical cauterization or when administered early after injury(12,27). This is probably the result of the drugs easy penetration into the scar area and of it reaching a maximum concentration during the second phase of stromal wound healing(27). To summarize, the antiangiogenic effects of bevacizumab appear to preferentially affect new, rather than established vessels(16).
Despite the fact that bevacizumab does not have direct anti-inflammatory effects and that its action during acute inflammation is not fully understood, we have confirmed that it is able to negatively affect fibroblast activity and decrease the number of inflammatory cells. Two high-affinity VEGF tyrosine kinase receptors have been described, kinase domain receptor (KDR) and fms-like tyrosine kinase (Flt-1). VEGF stimulates inflammation by modulating Flt-1 signaling(28). Recently, it has been shown that VEGF stimulates the proliferation of Tenon's fibroblasts and inhibits the proliferation of both human and rabbit fibroblasts with different concentrations of bevacizumab(29). The recruitment of monocytes and macrophages by VEGF-A plays a crucial role in inducing inflammatory neovascularisation(30). Anti-VEGF antibodies can bind VEGFR1 resulting in macrophage inhibition and neutrophil chemotaxis. Saravia et al.(31) reported that subconjunctival administration of bevacizumab abolished the inflammatory response, induced involution of new vessels, and resulted in the return of corneal function in herpes simplex virus type 1-infected rabbits. Furthermore, they demonstrated that a single subconjunctival injection of 10 μl of bevacizumab (25 μg/μl) leads to a significant decrease in neutrophils and blocks the inward flow of inflammatory cells into the stroma. This was reported to result from a VEGF-induced chemotaxis response by neutrophils and macrophages via the VEGFR1 receptor.
It is interesting to observe that the bevacizumab-induced inhibition of corneal neovascularization can occur within seven days. This suggests that the observed effects of bevacizumab might be increased by extending the duration of the study. Indeed, the main shortcomings of our experiment are the limited experiment time, along with the small sample size. Another limitation was the staining of sections using haematoxylin-eosin alone. In future studies, specific cell populations located in the cornea following cauterization can be detected using specific markers. For example, a good immunohistochemical marker for fibroblast activity is increased expression of the alpha smooth muscle actin protein.
CONCLUSION
Subconjunctival and topical administrations of bevacizumab both effectively inhibit corneal neovascularization, and decrease inflammation and fibroblast activity in a rat model of corneal neovascularization induced by alkali burn. Although the therapeutic concentration of bevacizumab that penetrated the cornea was not determined, we have demonstrated that a single subconjunctival dose of 0.05 ml (1.25 mg), or a twice daily topically applied bevacizumab solution of 10 mg/ml, is effective. Additional research is required to determine the optimal treatment schedules, dosage, intervals of application, and possible side-effects.